IGEM:Harvard/2006/Cyanobacteria/Notebook/2006-8-14: Difference between revisions
From OpenWetWare
Jump to navigationJump to search
JeffreyLau (talk | contribs) No edit summary |
JeffreyLau (talk | contribs) |
||
| Line 13: | Line 13: | ||
==To-do== | ==To-do== | ||
*'''Midiprep''' | *'''Midiprep''' | ||
*# Midiprep pSB4A3, J04500 (online instructions [http://www1.qiagen.com/literature/handbooks/PDF/PlasmidDNAPurification/PLS_Plasmid/1034637_HB_QIAGENPlasmid_112005.pdf here], page 16) | *# Midiprep pSB4A3, J04500 (online instructions [http://www1.qiagen.com/literature/handbooks/PDF/PlasmidDNAPurification/PLS_Plasmid/1034637_HB_QIAGENPlasmid_112005.pdf here], page 16) ''(in progress)'' | ||
*'''Insert:''' | *'''Insert:''' | ||
*#Digest miniprepped KaiA/B/C in GeneArt vector with X-P ''(in progress; 2 hr digest finishes at 7PM, 4 hr finishes at 9PM)'' | *#Digest miniprepped KaiA/B/C in GeneArt vector with X-P ''(in progress; 2 hr digest finishes at 7PM, 4 hr finishes at 9PM)'' | ||
| Line 31: | Line 31: | ||
*'''Western blots:''' | *'''Western blots:''' | ||
*#Ligate GFP dev\X-P and pSB4A3\S-P, transform ''(Wait until we've midiprepped more pSB4A3 and do another digest, because the pSB4A3\S-P from our digest assay was a curious size)'' | *#Ligate GFP dev\X-P and pSB4A3\S-P, transform ''(Wait until we've midiprepped more pSB4A3 and do another digest, because the pSB4A3\S-P from our digest assay was a curious size)'' | ||
*#Inoculate GFP dev + pSB1A2 | *#Inoculate GFP dev + pSB1A2 | ||
==E-gel images of colony PCR of J04500 + KaiA/B/C == | ==E-gel images of colony PCR of J04500 + KaiA/B/C == | ||
Revision as of 21:25, 14 August 2006
<html><style type='text/css'> .tabs {
font-size:80%;
font-weight:none;
width: 100%;
color: #FFFFFF;
background:#FFFFFF url("/images/5/54/DarkgreenTab-bg.gif") repeat-x bottom;
}
.tabs li {
background:url("/images/3/36/DarkgeenTab-left.gif") no-repeat left top;
}
.tabs a,.tabs strong {
background:url("/images/d/d3/DarkgreenTab-right.gif") no-repeat right top;
color:#FFFFFF;
padding: 3px 10px 3px 4px;
}
.tabs strong{
color:#CCFF00;
background-image:url("/images/b/b1/DarkgreenTab-right_on.gif");
}
.tabs a:hover{
color:#66FF00;
}
</style></html>
To-do
- Midiprep
- Midiprep pSB4A3, J04500 (online instructions here, page 16) (in progress)
- Insert:
- Digest miniprepped KaiA/B/C in GeneArt vector with X-P (in progress; 2 hr digest finishes at 7PM, 4 hr finishes at 9PM)
- Gel-purify
- (maybe) Inoculate more KaiA/B/C in GA vector
- Backbone:
- Digest J04500\S-P, \X-P (for Stage I constructs and BB high-copy) (in progress; 2 hr digest finishes at 7PM, 4 hr finishes at 9PM)
- CIP treatment of the above
- Gel-purify
Findand transform B0034
- KaiABC ligation:
- KaiABC\X-P + J04500\S-P (stage I)
- KaiABC\X-P + pSB1AK3 (well-formed BB) (we can purify this backbone from the J04500\X-P digest)
- KaiABC\X-P + B0034\S-P (stage I)
- Sequencing:
- Sequence miniprepped KaiA/B/C + GA vectors, natural KaiA
- Western blots:
- Ligate GFP dev\X-P and pSB4A3\S-P, transform (Wait until we've midiprepped more pSB4A3 and do another digest, because the pSB4A3\S-P from our digest assay was a curious size)
- Inoculate GFP dev + pSB1A2
E-gel images of colony PCR of J04500 + KaiA/B/C
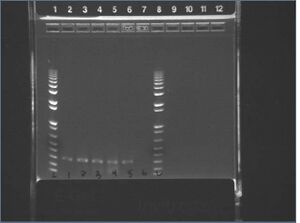
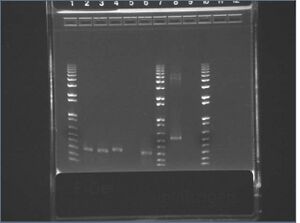
It appears that our vector self-ligated.
Digestion Assay for 8/14
| Label | DNA | Amount | Water | Enzymes | Amount | NEB buffer | BSA (100x) | Time | Master | Total | |
| Digests | 1 | J04500 | 19.5 µL | 1.75 µL | XbaI & PstI | 0.5 µL, 0.5 µL | Buffer 3 (2.5 µL) | 0.25 µL | 2 hrs | 1 | 25 µL |
| 2 | J04500 | 19.5 µL | 1.75 µL | SpeI & PstI | 0.5 µL, 0.5 µL | Buffer 2 (2.5 µL) | 0.25 µL | 2 hrs | 2 | 25 µL | |
| 3 | J04500 | 19.5 µL | 1.75 µL | XbaI & PstI | 0.5 µL, 0.5 µL | Buffer 3 (2.5 µL) | 0.25 µL | 4 hrs | 1 | 25 µL | |
| 4 | J04500 | 19.5 µL | 1.75 µL | SpeI & PstI | 0.5 µL, 0.5 µL | Buffer 2 (2.5 µL) | 0.25 µL | 4 hrs | 2 | 25 µL | |
| 5 | KaiA | 19.5 µL | 1.75 µL | XbaI & PstI | 0.5 µL, 0.5 µL | Buffer 3 (2.5 µL) | 0.25 µL | 2 hrs | 1 | 25 µL | |
| 6 | KaiB | 19.5 µL | 1.75 µL | XbaI & PstI | 0.5 µL, 0.5 µL | Buffer 3 (2.5 µL) | 0.25 µL | 2 hrs | 1 | 25 µL | |
| 7 | KaiC | 19.5 µL | 1.75 µL | XbaI & PstI | 0.5 µL, 0.5 µL | Buffer 3 (2.5 µL) | 0.25 µL | 2 hrs | 1 | 25 µL | |
| 8 | KaiA | 19.5 µL | 1.75 µL | XbaI & PstI | 0.5 µL, 0.5 µL | Buffer 3 (2.5 µL) | 0.25 µL | 4 hrs | 1 | 25 µL | |
| 9 | KaiB | 19.5 µL | 1.75 µL | XbaI & PstI | 0.5 µL, 0.5 µL | Buffer 3 (2.5 µL) | 0.25 µL | 4 hrs | 1 | 25 µL | |
| 10 | KaiC | 19.5 µL | 1.75 µL | XbaI & PstI | 0.5 µL, 0.5 µL | Buffer 3 (2.5 µL) | 0.25 µL | 4 hrs | 1 | 25 µL | |
| Master Mixes | 1 | - | - | 15.75 µL | XbaI & PstI | 4.5 µL, 4.5 µL | Buffer 3 (22.5 µL) | 2.25 µL | - | - | 49.5 µL |
| 2 | - | - | 7 µL | SpeI & PstI | 2 µL, 2 µL | Buffer 2 (10 µL) | 1 µL | - | - | 22 µL |
Add 5.5 µL of the corresponding master to each tube.