CH391L/S12/Artemisinic Acid Engineering: Difference between revisions
No edit summary |
No edit summary |
||
| Line 13: | Line 13: | ||
Aretmisinin, or Qinghaosu, is a chemical compound produced naturally by the plant <i>Artemsia annua</i>. It is a sesquiterpene lactone, a type of terpene derived from 3 isoprene units and a lactone ring. The compound is unique, as it contains an oxygenated cyclic ring with a peroxide bridge, and contains no Nitrogen atoms. It was first derived from <i>Artemsia annua</i> in 1971 by Chinese chemists interested in observing the plant's supposed use in curing headaches <cite>Klayman1985</cite>. In response to the Chinese government's push to develop new malaria cures, malaria patients were treated with this compound<cite>Brown2010</cite>. Fortunately, it was determined to be very effective at combating common malaria strains. | Aretmisinin, or Qinghaosu, is a chemical compound produced naturally by the plant <i>Artemsia annua</i>. It is a sesquiterpene lactone, a type of terpene derived from 3 isoprene units and a lactone ring. The compound is unique, as it contains an oxygenated cyclic ring with a peroxide bridge, and contains no Nitrogen atoms. It was first derived from <i>Artemsia annua</i> in 1971 by Chinese chemists interested in observing the plant's supposed use in curing headaches <cite>Klayman1985</cite>. In response to the Chinese government's push to develop new malaria cures, malaria patients were treated with this compound<cite>Brown2010</cite>. Fortunately, it was determined to be very effective at combating common malaria strains. | ||
Artemisinin posses antimalarial capabilities due to its trioxane ring structure. It is capable of killing the malaria parasite while infecting human red blood cells, and even proved effective on strains resistant to commonly used antimalarial drugs. Unfortunately, the | Artemisinin posses antimalarial capabilities due to its trioxane ring structure. It is capable of killing the malaria parasite while infecting human red blood cells, and even proved effective on strains resistant to commonly used antimalarial drugs. Unfortunately, the ketone group on the bottom of artemisinin, shown below, imparts negative characteristics. In its native form, artemisinin has low solubility in both water and oil. This makes the compound difficult to produce and distribute, and led to the development of its multiple derivatives shown below. Note the expansion of the ketone group on the bottom of artemisinin in successive derivatives, which contribute to their increased solubility and potency<cite>Brown2010</cite>. | ||
[[Image:ArtemisininDerivates.jpg]] | [[Image:ArtemisininDerivates.jpg]] | ||
| Line 19: | Line 19: | ||
==Chloroqunie== | ==Chloroqunie== | ||
Previously, one of the most widely used antimalarial drugs produced was chloroquine. While successful at combating malaria, chloroquine exhibits multiple adverse side effects, most notably Pruritus. Pruritus is a particularly | Previously, one of the most widely used antimalarial drugs produced was chloroquine. While successful at combating malaria, chloroquine exhibits multiple adverse side effects, most notably Pruritus. Pruritus is a particularly uncomfortable form of itching sensation that dark-skinned individuals are more susceptible to, due to their increased melanocytes and the pathway in which chloroquine binds to these melanocytes<cite>Aghahowa2010</cite>. This condition can be so painful that multiple drugs must be administered in unison with chloroquine in order to negate its detrimental side-effects. Considering that malaria generally plagues the poorest 20% of the world's population, mainly located in countries in Africa, the cost-effectiveness of this procedure can make it impractical to implement. | ||
==Production of Artemisinic Acid== | ==Production of Artemisinic Acid== | ||
| Line 25: | Line 25: | ||
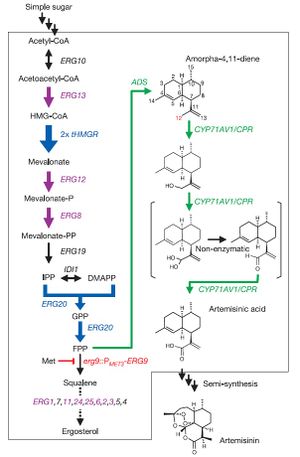
[[Image:ArtemisnicAcidPathway.jpg|thumb|left|Ro et al.'s procedure for converting carbohydrates into artemisinic acid in <i>S. cerevisiae</i>. <cite>Ro2006</cite>.]] | [[Image:ArtemisnicAcidPathway.jpg|thumb|left|Ro et al.'s procedure for converting carbohydrates into artemisinic acid in <i>S. cerevisiae</i>. <cite>Ro2006</cite>.]] | ||
In a famous paper released by Ro et al., <i> Saccharomyces cerevisiae</i> was engineered to produce artemisinic acid from simple sugars. This involved metabolic engineering of the mevalonate pathway, | In a famous paper released by Ro et al., <i> Saccharomyces cerevisiae</i> was engineered to produce artemisinic acid from simple sugars. This involved metabolic engineering of the mevalonate pathway, naturally present in <i>S. cerevisiae</i>, to introduce a three-step pathway that converts amorpha-4,11-diene into artemisinic acid. | ||
The first step of this process involved engineering the yeasts' natural mevalonate pathway to produce increased levels of farnesyl pyrophosphate (FPP). 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase (tHMGR) was | The first step of this process involved engineering the yeasts' natural mevalonate pathway to produce increased levels of farnesyl pyrophosphate (FPP). 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase (tHMGR) was over-expressed in <i>S. cerevisiae</i> by introducing nother copy of the gene encoding for this enzyme, leading to increased mevalonate production by fivefold. Additionally, the mutant allele <i>upc2-1</i> was introduced, leading to moderate over-expression of various ERG genes responsible for the production of enzymes in the mevalonate pathway. | ||
<i>S. cerevisiae</i> naturally employes mevalonate to produce sterols. Ro et al. therefore introduced a methionine-repressible promoter to down-regulate the enzyme ERG9, responsible for the conversion of | <i>S. cerevisiae</i> naturally employes mevalonate to produce sterols. Ro et al. therefore introduced a methionine-repressible promoter to down-regulate the enzyme-encoding gene ERG9, responsible for the conversion of mevalonate into squalene. | ||
It is worthwhile to note that the process by which artemisinic acid is converted into artemisinin is currently under debate, as "...how artemisinic acid transforms into artemisinin is still under investigation" <cite>Wen2011</cite>. Much of the work accomplished by Ro et al. has shed light on the mevalonate pathway and its relation to the production of | Natively, <i>S. cerevisiae</i> contains no pathway to produce any form of artemisinin or its precursors. Therefore, Ro et al. isolated the genes encoding amorphadiene synthase (ADS) from <i>Artemsia annua</i>, and introduced them into <i>S. cerevisiae</i>. | ||
It is worthwhile to note that the process by which artemisinic acid is converted into artemisinin is currently under debate, as "...how artemisinic acid transforms into artemisinin is still under investigation" <cite>Wen2011</cite>. Much of the work accomplished by Ro et al. has shed light on the mevalonate pathway and its relation to the production of artemisinic acid. | |||
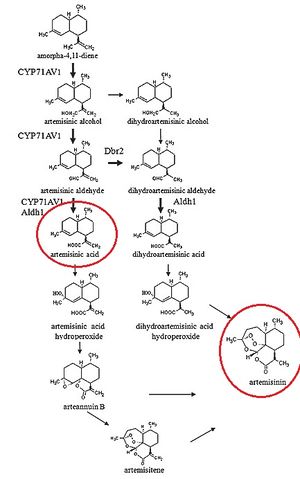
[[Image:BiosyntheticArtemisininProduction.jpg|thumb|right|Proposed mechanism for the production of artemisinin in <i>Artemsia annua</i>. <cite>Wen2011</cite>.]] | [[Image:BiosyntheticArtemisininProduction.jpg|thumb|right|Proposed mechanism for the production of artemisinin in <i>Artemsia annua</i>. <cite>Wen2011</cite>.]] | ||
Revision as of 17:07, 8 April 2012
- NOTE* Page is still under construction. As long as this note appears, assume information presented on this page is incomplete and unreliable.
Artemisia

Artemisia is a genus that encompass a broad range of plants commonly known as sagebrush, sagewort, and wormwood. The family, which derives its name from the either from the Greek goddess Artemis or Artemisia, the wife of Mausolus, includes many famous plants. Artemisia absinthium is used in the production of Absinthe, and Shakespeare employed wormwood as a cure for the love potion in Midsummer's Night Dream. One species of Artemisia, known as Artemsia annua, has been used for thousands of years in traditional Chinese medicines to cure headaches.
This last species is of particular interest to synthetic biologists, due to its ability to produce Artemisinin. As a result, much of the species' genes have been characterized and submitted to the Gene Bank.
Artemisinin
Aretmisinin, or Qinghaosu, is a chemical compound produced naturally by the plant Artemsia annua. It is a sesquiterpene lactone, a type of terpene derived from 3 isoprene units and a lactone ring. The compound is unique, as it contains an oxygenated cyclic ring with a peroxide bridge, and contains no Nitrogen atoms. It was first derived from Artemsia annua in 1971 by Chinese chemists interested in observing the plant's supposed use in curing headaches [1]. In response to the Chinese government's push to develop new malaria cures, malaria patients were treated with this compound[2]. Fortunately, it was determined to be very effective at combating common malaria strains.
Artemisinin posses antimalarial capabilities due to its trioxane ring structure. It is capable of killing the malaria parasite while infecting human red blood cells, and even proved effective on strains resistant to commonly used antimalarial drugs. Unfortunately, the ketone group on the bottom of artemisinin, shown below, imparts negative characteristics. In its native form, artemisinin has low solubility in both water and oil. This makes the compound difficult to produce and distribute, and led to the development of its multiple derivatives shown below. Note the expansion of the ketone group on the bottom of artemisinin in successive derivatives, which contribute to their increased solubility and potency[2].
Chloroqunie
Previously, one of the most widely used antimalarial drugs produced was chloroquine. While successful at combating malaria, chloroquine exhibits multiple adverse side effects, most notably Pruritus. Pruritus is a particularly uncomfortable form of itching sensation that dark-skinned individuals are more susceptible to, due to their increased melanocytes and the pathway in which chloroquine binds to these melanocytes[3]. This condition can be so painful that multiple drugs must be administered in unison with chloroquine in order to negate its detrimental side-effects. Considering that malaria generally plagues the poorest 20% of the world's population, mainly located in countries in Africa, the cost-effectiveness of this procedure can make it impractical to implement.
Production of Artemisinic Acid
In a famous paper released by Ro et al., Saccharomyces cerevisiae was engineered to produce artemisinic acid from simple sugars. This involved metabolic engineering of the mevalonate pathway, naturally present in S. cerevisiae, to introduce a three-step pathway that converts amorpha-4,11-diene into artemisinic acid.
The first step of this process involved engineering the yeasts' natural mevalonate pathway to produce increased levels of farnesyl pyrophosphate (FPP). 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase (tHMGR) was over-expressed in S. cerevisiae by introducing nother copy of the gene encoding for this enzyme, leading to increased mevalonate production by fivefold. Additionally, the mutant allele upc2-1 was introduced, leading to moderate over-expression of various ERG genes responsible for the production of enzymes in the mevalonate pathway.
S. cerevisiae naturally employes mevalonate to produce sterols. Ro et al. therefore introduced a methionine-repressible promoter to down-regulate the enzyme-encoding gene ERG9, responsible for the conversion of mevalonate into squalene.
Natively, S. cerevisiae contains no pathway to produce any form of artemisinin or its precursors. Therefore, Ro et al. isolated the genes encoding amorphadiene synthase (ADS) from Artemsia annua, and introduced them into S. cerevisiae.
It is worthwhile to note that the process by which artemisinic acid is converted into artemisinin is currently under debate, as "...how artemisinic acid transforms into artemisinin is still under investigation" [5]. Much of the work accomplished by Ro et al. has shed light on the mevalonate pathway and its relation to the production of artemisinic acid.
Relation to iGEM
There has been no incidence of previous iGEM teams investigating artemisinin or any of its precursors. Many iGEM teams have referenced Ro et al., but only as an example of pathway engineering, or in order to discuss ethics and synthetic biology's potential to positively benefit society. No parts in the parts registry deals with artemisinin either. Only one [set of parts] mentions a protein, Granulysin, which can potentially be used to fight malaria.
References
- Klayman DL. Qinghaosu (artemisinin): an antimalarial drug from China. Science. 1985 May 31;228(4703):1049-55. DOI:10.1126/science.3887571 |
- Brown GD. The biosynthesis of artemisinin (Qinghaosu) and the phytochemistry of Artemisia annua L. (Qinghao). Molecules. 2010 Oct 28;15(11):7603-98. DOI:10.3390/molecules15117603 |
- Aghahowa SE, Obianwu HO, Isah AO, and Arhewoh IM. Chloroquine-induced Pruritus. Indian J Pharm Sci. 2010 May;72(3):283-9. DOI:10.4103/0250-474X.70471 |
- Ro DK, Paradise EM, Ouellet M, Fisher KJ, Newman KL, Ndungu JM, Ho KA, Eachus RA, Ham TS, Kirby J, Chang MC, Withers ST, Shiba Y, Sarpong R, and Keasling JD. Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature. 2006 Apr 13;440(7086):940-3. DOI:10.1038/nature04640 |
- Wen W and Yu R. Artemisinin biosynthesis and its regulatory enzymes: Progress and perspective. Pharmacogn Rev. 2011 Jul;5(10):189-94. DOI:10.4103/0973-7847.91118 |
