IGEM:Harvard/2009/Notebook/Harvard iGEM 2010/2010/07/06: Difference between revisions
| Line 120: | Line 120: | ||
Gel #1 | Gel #1 | ||
-Lane 1: 1kb Ladder | |||
-Lanes 2-4: Ger Sense | |||
-Lanes 5-7: Ger Antisense | |||
-Lanes 8-10: LTP Sense | |||
-Lanes 11-13: LTP Antisense | |||
-Lanes 14-16: Bet 2 Sense | |||
-Lanes 17-19: Bet 2 Antisense | |||
-Lanes 20-22: Bet 1 Sense | |||
-Lanes 23-25: Bet 1 Antisense | |||
Revision as of 09:31, 7 July 2010
 iGEM iGarden iGEM iGarden
|
<html><img src="/images/9/94/Report.png" border="0" /></html> Main project page <html><img src="/images/c/c3/Resultset_previous.png" border="0" /></html>Previous entry<html> </html>Next entry<html><img src="/images/5/5c/Resultset_next.png" border="0" /></html> | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Team AllergyToday, we isolated plasmid that contained antisense and sense sequences of the allergen panel and introns PME and PAL that we grew last week. ProceduresFor amiRNA
For ihpRNA
ResultsFor amiRNAMiniprep of turbo bacteria for plasmids containing sequences for amiRNA Should obtain plasmids containing the complete sequence for amiRNA. The resulting concentration should be relatively high (<100ng/μL) because all growing bacteria would have the plasmids inside. Nanodrop of miniprepped amiRNA plasmids We used 2μL of DNA per nanodrop.
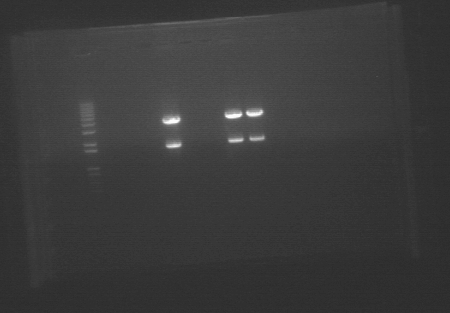
Diagnostic digest of amiRNA plasmids Digested 200~500ng of plasmids with EcoR1 and Spe1. We will then run the digested vectors on an E Gel and look for DNA that is the length of the insert. If such DNA appears on the gel, then we will send it off to be sequenced. E Gel of digested amiRNA  We had to run 21 samples, so we used two gels. The orders were: Gel #1
Gel #2
For ihpRNAMiniprep of plasmids containing introns for hpRNA Nanodrop of miniprepped intron plasmids We used 2μL of DNA per nanodrop.
Gel #1 -Lane 1: 1kb Ladder -Lanes 2-4: Ger Sense -Lanes 5-7: Ger Antisense -Lanes 8-10: LTP Sense -Lanes 11-13: LTP Antisense -Lanes 14-16: Bet 2 Sense -Lanes 17-19: Bet 2 Antisense -Lanes 20-22: Bet 1 Sense -Lanes 23-25: Bet 1 Antisense
Team FenceInoculated Barstar and NLS colonies from Thursday's ligation. Performed PCR on LacIn (E10) following standard PCR measurements as previously described. Digestion of B21 backbone using Xba1 and Pst1 Total volume per reaction: 50 μL
Digestion successful, larger bands removed for gel extraction. Gel extraction performed according to QIAgen gel extraction protocol. Team FlavourTagging Miraculin and Brazzein with YFP and StrepII tagsTag: -E--N--X--STREP/YFP--S--N--P-
GelThe gel showed DNA fragments consistent with Miraculin and Brazzein. The digestion of B21 appeared to be succesful, but the DNA sequence cut out was too small to see on the gel. Gel Lanes:
1. 1 kb plus ladder
2. B21 speI/pstI
4. B21 ecoRI/xbaI
6. Miraculin xbaI/pstI
8. Miraculin ecoRI/speI
10. Brazzein xbaI/pstI
12. Brazzein ecoRI/pstI
14. 1kb plus ladder
ODs of gel purified DNA
B21 speI/pstI: 15.9 ng/μL
B21 ecoRI/xbaI: 11.0 ng/μL
Miraculin xbaI/pstI: 9.7 ng/μL
Miraculin ecoRI/speI: 7.1 ng/μL
Brazzein xbaI/pstI: 2.6 ng/μL
Brazzein ecoRI/pstI: 5.0 ng/μL
LigationWe did 6 different ligation reactions. The chart below shows the different reactions. Ligation reactions were left at room temperature for 15 minutes.
TransformationWe mixed 5μL ligation mix with 15μL Turbo e. coli cells. These were placed in ice for 30 minutes and then heat shocked in 42°C water bath for 30 seconds. The cells were placed back on ice for 2 minutes. 170 μL of SOC broth was added to each Eppendorf tube. The transformed e. coli were then plated on LB Ampicillin plates and left in the 37°C incubator overnight. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||