User:Alicia Rasines Mazo/Notebook/CHEM-581 Experimental Chemistry I/2014/09/10: Difference between revisions
From OpenWetWare
No edit summary |
No edit summary |
||
| (8 intermediate revisions by the same user not shown) | |||
| Line 22: | Line 22: | ||
===DSC (Differential Scanning Calorimetry) Data=== | ===DSC (Differential Scanning Calorimetry) Data=== | ||
Note: To calculate mass of water evaporated the following equation was used: q=mHv. So that mass of water=(q×mass of sample)/Hv where Hv=2.23×10<sup>3</sup> J/g | Note: To calculate mass of water evaporated the following equation was used: q=mHv. So that mass of water=(q×mass of sample)/Hv where Hv=2.23×10<sup>3</sup> J/g<br.> | ||
'''PVA film in 2 ppm MG solution'''<br.> | |||
[[Image:PVA2ppmDSC.png]]<br.> | |||
* Onset of water evaporation: 99.59ºC | |||
* Heat of hydrate decomposition: 830.4 J/g | |||
* Mass of water evaporated = 0.00144 g | |||
* Major peaks in water: | |||
** Minimum at 134.73ºC, other peaks at 101, 103 ºC | |||
* Glass transition: | |||
** Top curve inflection point: 68.58ºC | |||
** Bottom curve inflection points: 69.39, 85.00ºC<br.> | |||
'''PVA film in 8 ppm MG solution'''<br.> | |||
[[Image:PVA8ppmDSC.png]] | |||
* Onset of water evaporation: 100.91ºC | |||
* Heat of hydrate decomposition: 1130 J/g | |||
* Mass of water evaporated = 0.00177 g | |||
* Major peaks in water: | |||
** Minimum at 103.58ºC, other peaks at 107, 117 and 120ºC | |||
* Glass transition: | |||
** Top curve inflection point: 68.11ºC | |||
** Bottom curve inflection points: 68.94, 87.66 ºC<br.> | |||
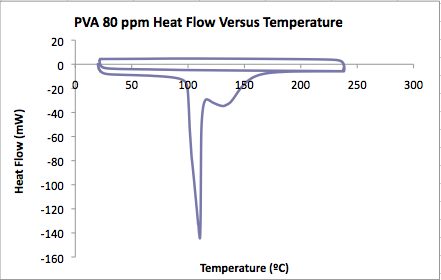
'''PVA film in 80 ppm MG solution'''<br.> | |||
[[Image:PVA80ppmDSC.png]] | |||
* Onset of water evaporation: 99.82ºC | |||
* Heat of hydrate decomposition: 1458 J/g | |||
* Mass of water evaporated = 0.00257 g | |||
* Major peaks in water: | |||
** Minimum at 110ºC, other peaks at 117 and 130. | |||
* Glass transition: | |||
** Top curve inflection point: 68.17ºC | |||
** Bottom curve inflection point: 68.56ºC<br.> | |||
'''PVA film in 200 ppm MG solution'''<br.> | |||
[[Image:PVA200ppmDSC.png]] | |||
* Onset of water evaporation: 99.77ºC | |||
* Heat of hydrate decomposition: 938.2 J/g | |||
* Mass of water evaporated = 0.00147 g | |||
* Major peaks in water: | |||
** Minimum at 125.54ºC, other peaks at 101, 105 and 136 | |||
* Glass transition: | |||
** Top curve inflection point: 68.36ºC | |||
** Bottom curve inflection point: 69.15 ºC<br.> | |||
'''PVA-Clay film in 2 ppm MG solution'''<br.> | |||
[[Image:PVAClay2ppmDSC.png]] | |||
* Onset of water evaporation: 100.51ºC | |||
* Heat of hydrate decomposition: 1276 J/g | |||
* Mass of water evaporated = 0.00232 g | |||
* Major peaks in water: | |||
** Minimum at 107ºC, other peaks at 109, 111 and 125ºC | |||
* Glass transition: | |||
** Top curve inflection point: 68.16ºC | |||
** Bottom curve inflection point: 68.70 ºC<br.> | |||
'''PVA-Clay film in 8 ppm MG solution'''<br.> | |||
[[Image:PVAClay80ppmDSC.png]] | |||
* Onset of water evaporation: 98.55ºC | |||
* Heat of hydrate decomposition: 865.9 J/g | |||
* Mass of water evaporated = 0.00213 g | |||
* Major peaks in water: | |||
** Minimum at 101.19ºC, other peaks at 104, 130ºC | |||
* Glass transition: | |||
** Top curve inflection point: 68.59ºC | |||
** Bottom curve inflection point: 69.02 ºC<br.> | |||
'''PVA-Clay film in 80 ppm MG solution'''<br.> | |||
[[Image:PVAClay80ppm2DSC.png]] | |||
* Onset of water evaporation: 100.76ºC | |||
* Heat of hydrate decomposition: 1044 J/g | |||
* Mass of water evaporated = 0.00158 g | |||
* Major peaks in water: | |||
** Minimum at 101.94ºC, other peaks at 105, 108ºC | |||
* Glass transition: | |||
** Top curve inflection point: 68.27ºC | |||
** Bottom curve inflection point: 68.50 ºC<br.> | |||
'''PVA-Clay film in 200 ppm MG solution'''<br.> | |||
[[Image:PVACLay200ppm.png]]<br.> | |||
* Onset of water evaporation: 99.70ºC | |||
* Heat of hydrate decomposition: 1138 J/g | |||
* Mass of water evaporated = 0.00177 g | |||
* Major peaks in water: | |||
** Minimum at 108ºC, other peaks at 110, 131ºC | |||
* Glass transition: | |||
** Top curve inflection point: 68.54ºC | |||
** Bottom curve inflection point: 67.97 ºC | |||
<!-- ##### DO NOT edit below this line unless you know what you are doing. ##### --> | <!-- ##### DO NOT edit below this line unless you know what you are doing. ##### --> | ||
|} | |} | ||
__NOTOC__ | __NOTOC__ | ||
Revision as of 09:02, 24 September 2014
| <html><img src="/images/9/94/Report.png" border="0" /></html> Main project page <html><img src="/images/c/c3/Resultset_previous.png" border="0" /></html>Previous entry<html> </html>Next entry<html><img src="/images/5/5c/Resultset_next.png" border="0" /></html> | |
Tasks for September 10
Completion of Film SynthesisContinued from Dr. Harting's procedure
Film preparation of ionic liquid modified clay
DSC (Differential Scanning Calorimetry) DataNote: To calculate mass of water evaporated the following equation was used: q=mHv. So that mass of water=(q×mass of sample)/Hv where Hv=2.23×103 J/g<br.>
PVA film in 2 ppm MG solution<br.>
PVA film in 8 ppm MG solution<br.>
PVA film in 80 ppm MG solution<br.>
PVA film in 200 ppm MG solution<br.>
PVA-Clay film in 2 ppm MG solution<br.>
PVA-Clay film in 8 ppm MG solution<br.>
PVA-Clay film in 80 ppm MG solution<br.>
PVA-Clay film in 200 ppm MG solution<br.>
| |