User:Floriane Briere/Notebook/CHEM-496/2011/09/14: Difference between revisions
No edit summary |
|||
| (17 intermediate revisions by 2 users not shown) | |||
| Line 11: | Line 11: | ||
==Objective== | ==Objective== | ||
Today's experiment | Today's experiment consists on performing a Bradford Assay technique which is going to allow us to determine the unknown proteins’ concentration of a solution. We are going to use 6 standards solutions of BSA whose concentrations are known. Then, thanks to a spectrophotometer, we’ll be able to determine the concentration of the MBP solution. | ||
==Protocol== | ==Protocol== | ||
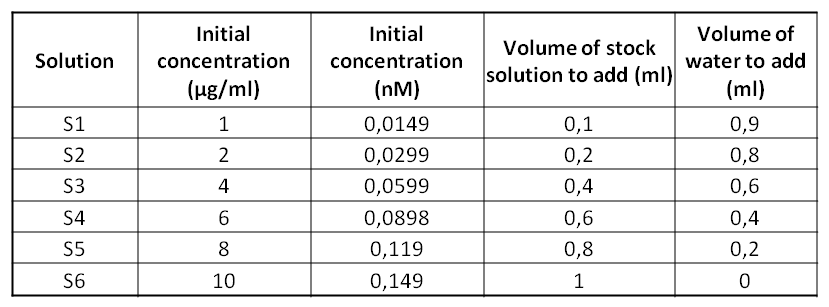
*Preparation of the stock solution (4ml - 10µg/ml) | *Preparation of the stock solution (4ml - 10µg/ml) | ||
#Add 0.027ml of BSA (1.46mg/ml) to 3.973ml of water | #Add 0.027ml of BSA (1.46mg/ml - 66776 g/mol) to 3.973ml of water | ||
#Vortex the solution | #Vortex the solution | ||
| Line 33: | Line 33: | ||
# Add 1ml of water to 200µl of Bradford reagent and measure absorbance at 595nm (blank) | # Add 1ml of water to 200µl of Bradford reagent and measure absorbance at 595nm (blank) | ||
# Measure absorbance at 595nm of only MPB diluted solution (1/100 and 1/1000) | # Measure absorbance at 595nm of only MPB diluted solution (1/100 and 1/1000) | ||
[[Image:14sept - Tableau SSolutionN2.png]] | |||
==Results== | ==Results== | ||
* Curve of the concentration (nM) as a function of the Absorbance at 595nm: | |||
[[Image:14sept - Abs(595=f(concentration).png]] | [[Image:14sept - Abs(595)=f(concentration).png]] | ||
To draw this curve, we add to take into account that the Bradford Assay was made with 5/6 diluted solutions (because we added 1ml of solution to 200µl of Bradford reagent). | To draw this curve, we add to take into account that the Bradford Assay was made with 5/6 diluted solutions (because we added 1ml of solution to 200µl of Bradford reagent). | ||
Red lines represent the Absorbance at 595nm of the 5/6000 diluted MPB solution. This curve allow us to determine the concentration of the MPB solution. | |||
We can calculate the unknown concentration thanks to the line’s equation (y = 2.1105x - 0.0204): | |||
'''* Curve of the Absorbance as a function of the Wavelength (nm) | Abs(595nm) = 2.1105 * concentration - 0.0204 | ||
=> concentration = (0.44 + 0.0204)/2.1105 = 0.218nM | |||
So, the 5/6 diluted MPB solution has a concentration of 0.218nM. | |||
The pure MPB solution has a concentration of '''0.261nM (0.218*(6/5) = 0.261nM)'''. | |||
<b>What are the final concentrations (in μg/mL of your standard solutions? Are they diluted from your original calculations. Your value for the concentration of MBP seems a bit low from where it should be. Also, you needed to take a spectrum of just MBP in water/buffer with NO Bradford Assay reagent. We need that so we can calculate ε.</b> [[User:Matt Hartings|Matt Hartings]] 20:47, 20 September 2011 (EDT) | |||
* Curve of the Absorbance as a function of the Wavelength (nm): | |||
[[Image:14sept - Absorbance = f(wavelength).png]] | |||
This curve is made with the standards solution and the 1/1000 diluted MPB solution (with bradford reagent)) measures. | |||
[[Image:14sept - Absorbance = f(wavelength) v2.png]] | [[Image:14sept - Absorbance = f(wavelength) v2.png]] | ||
This curve is made with the 1/1000 diluted MPB solution measures. | This curve is made with the 1/1000 diluted MPB solution (without Bradford reagent) measures. | ||
* Curve of the molar absorptivity (L.mol^-1.cm^-1) as a function of the Wavelength (nm): | |||
= | [[Image:14sept - Molar absorbitivity= f(wavelength).png]] | ||
According to Beer-Lambert law, Absorbance = molar absorptivity (L.mol^-1.cm^-1) * concentration (M) * length of the cuve (cm) | |||
So, molar absorptivity (ε) = Absorbance/(concentration (c)* length of the cuve (l)) | |||
We used the Absorbance of the 1/1000 MPB solution (without Bradford reagent) whose concentration is 29,16*10^-12M. And we know that l = 1cm | |||
Revision as of 18:09, 8 December 2011
| File:BDLlogo notext ir.png Project name | <html><img src="/images/9/94/Report.png" border="0" /></html> Main project page <html><img src="/images/c/c3/Resultset_previous.png" border="0" /></html>Previous entry<html> </html>Next entry<html><img src="/images/5/5c/Resultset_next.png" border="0" /></html> |
|
ObjectiveToday's experiment consists on performing a Bradford Assay technique which is going to allow us to determine the unknown proteins’ concentration of a solution. We are going to use 6 standards solutions of BSA whose concentrations are known. Then, thanks to a spectrophotometer, we’ll be able to determine the concentration of the MBP solution. Protocol
Results
To draw this curve, we add to take into account that the Bradford Assay was made with 5/6 diluted solutions (because we added 1ml of solution to 200µl of Bradford reagent). Red lines represent the Absorbance at 595nm of the 5/6000 diluted MPB solution. This curve allow us to determine the concentration of the MPB solution. We can calculate the unknown concentration thanks to the line’s equation (y = 2.1105x - 0.0204): Abs(595nm) = 2.1105 * concentration - 0.0204 => concentration = (0.44 + 0.0204)/2.1105 = 0.218nM So, the 5/6 diluted MPB solution has a concentration of 0.218nM. The pure MPB solution has a concentration of 0.261nM (0.218*(6/5) = 0.261nM). What are the final concentrations (in μg/mL of your standard solutions? Are they diluted from your original calculations. Your value for the concentration of MBP seems a bit low from where it should be. Also, you needed to take a spectrum of just MBP in water/buffer with NO Bradford Assay reagent. We need that so we can calculate ε. Matt Hartings 20:47, 20 September 2011 (EDT)
This curve is made with the standards solution and the 1/1000 diluted MPB solution (with bradford reagent)) measures. This curve is made with the 1/1000 diluted MPB solution (without Bradford reagent) measures.
According to Beer-Lambert law, Absorbance = molar absorptivity (L.mol^-1.cm^-1) * concentration (M) * length of the cuve (cm) So, molar absorptivity (ε) = Absorbance/(concentration (c)* length of the cuve (l)) We used the Absorbance of the 1/1000 MPB solution (without Bradford reagent) whose concentration is 29,16*10^-12M. And we know that l = 1cm
| |