User:Trisha I. Ibeh/Notebook/Trisha Notebook/2013/09/03
 Biomaterials Design Lab Biomaterials Design Lab
|
<html><img src="/images/9/94/Report.png" border="0" /></html> Main project page <html><img src="/images/c/c3/Resultset_previous.png" border="0" /></html>Previous entry<html> </html>Next entry<html><img src="/images/5/5c/Resultset_next.png" border="0" /></html> | ||||||||||||||||
|
The template for this lab can be seen from Dr. Hartings lab. Values are altered to accurately describe the lab that was conducted on this day. The template can be found here ObjectiveThe molar absorptivities of two different molecules, adenosine and inosine were determined in this lab using UV-Vis and Beer's law. The changes in UV-Vis spectra will be observed to determine changes in concentration of both adenosine and inosine. In order to do this, the molar absorptivity (ε) of both of these molecules will be known. A calibration curve from the class data will be created. From this data the standard deviation, Confidence Interval (90% and 95% confidence) will be calculated and Grubb's test will be performed to determine the outlier.
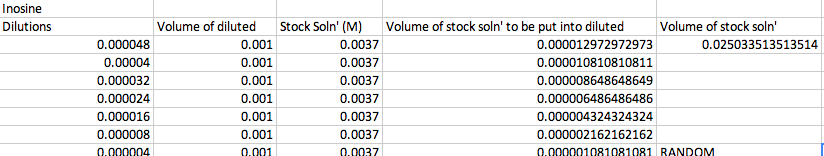
DilutionsStock Solutions Stock solutions were made to create these dilutions for each molecule. The calculations for the stock solution and dilutions were performed before lab.
The RANDOM concentration was 0.25*10^-5 for adenosine and 0.4*10^-5 for inosine. Preparation of Dilutions of Adenosine Preparation of Dilutions of Inosine
DataAdenosine Absorbance Spectrum Adenosine Calibration Curve NotesDue to the time restrictions of the lab, the inosine spectra was taken the next lab (9/4/13).
| |||||||||||||||||