User:David J Weiss/Notebook/notes Photoelect
Photoelectric Effect Notes
SJK 17:27, 25 October 2009 (EDT)

Your primary lab notebook up until the point of analysis is excellent. Very good description of equipment and raw data spreadsheets. You do a good job putting in the analysis equations and graphs...however, the analysis commentary is missing and / or doesn't seem thoughtful. So, that was a big issue. I don't think you came close to getting the best analysis out of your data.
In this lab we will examine the effects of the photoelectric effect and try to determine Plank's Constant.
Need More
Equipment
- Mercury Vapor Light Source and Light Block(Model OS-9286)
- Voltage Range: 108-132 VAC
- Power: 125 W MAX
- Frequency: 47-63 HZ
- 115 Volts
- Light Aperture (Model AP-9369)
- Coupling Bar (Model AP-9369)
- Digital Voltmeter (Model 37XR-A)and/or (model FLUKE 111)
- 2 Connection Cables (Model 8-24)
- Stopwatch
- Two 9 Volt Batteries
- Duracell-Procell
- h/e Apparatus (Model AP-9369)
- 3 Filters
- Relative Transmission
- Yellow Line
- Green Line
Set Up

- First turn on the lamp and let it warm up for 20 minutes as per instructions on instrument
- Connect the voltmeter to the h/e apparatus and check the battery (15.9V) and then connect the voltmeter to the h/e appratus
- Position the lamp and the h/e apparatus so that the emitted light shines on the opening of the h/e apparatus
- Focus the beam by adjusting the light aperture on the adjusting rods
Safety
The major safety concerns in this lab are:
- Electrical avoid shocks from the Lamp
- Avoid damaging the equipment
- Avoid turning the lamp on and off due to potential damage to the bulb
- Avoid touching the filters due to fingerprints interfering with the filters
- Toxic Chemicals from the mercury lamp
Notes
Day 1
- Worked with Elizabeth Allen.
- We first had the Safety brief with Dr Koch. After that we then turned the lamp on so that it could warm up for 20 minutes. After we then aligned and set up the equipment. We then measured the voltage of the h/e apparatus using the multimeter which was 15.9V. We then adjusted the focus of the light in relation to the h/e apparatus.
Experiment 1: The Photon Theory of Light
- Started with the experiment by first recording the time it takes to charge and what the charge is, starting with the ultra violet then the violet then the blue then the green and finally yellow. For each color we went through each filter 100%, 80%, 60%, 40%, and 20%. For the yellow and green we also had to add another filter corresponding to the color.
Experiment 2: Determination of h
We did not have enough time to complete this part of the experiment due to time running out.
Day 2
- Worked with Elizabeth Allen
- We came into lab and set up the equipment again and turned on the lamp and let it warm up for 20 min again.
- We changed out the battery's on the h/e apparatus and a new voltage of 18.9 V was read on the multimeter. Due to the reason that the violet light was taking a while to reach a charge and that it was not reaching the same voltage as from the day before.
- We talked with Dr Koch and could not still figure out why it was happening so we deiced to just move on to the second experiment and come in on Wednesday to repeat the experiment with more success.
Experiment 1
- Started with the experiment by first recording the time it takes to charge and what the charge is, starting with the ultra violet then the violet then the blue then the green and finally yellow. For each color we went through each filter 100%, 80%, 60%, 40%, and 20%.
- Having some difficulty in obtaining the same values for the voltages while doing the first experiment. The voltage is off by a factor of about .05 volts i cannot see the reason why. The time required for it to charge is also taking a longer time to fully charge the reason why is still unknown. Got some data but got frustrated could not figure out why the data kept differing with each try.
Experiment 2
- Started with the first order colors and took 3 measurements of each starting with violet and then going to blue green and yellow, both the green and yellow had corresponding filters.
- After we took the first order readings we then moved on to the 2nd order reading starting again with the violet and again down the line with the same order adding the filters to the green and yellow respectfully. We had to readjust the focus when we moved to the 2nd order due to the fact that we had to change the distance in between the light and the detector to be able to view the 2nd order color spectrum. We also had to make some adjustments between the different colors in the 2nd order between the blue and the green/yellow spectral lines.
Day 3
- Worked with Elizabeth Allen
- We deiced to come into lab again to repeat the first part of the experiment due to the lack of data on day 2, and get a new set of data for experiment 2 also due to the lack of data from day one and running out of time in day 1.
- We came in and set up the equipment, warmed up the lamp adjusted the focus on the lamp turned on the h/e apparatus and plugged in the multimeter and took the voltage on the h/e apparatus which was 18.9 V.
Experiment 1
- This time we deiced to start with the ultra violet light and then through the same colors we used the first two days.
- We again ran into some of the same problems again in that the reading were somewhat erratic in the voltages but we overcame these and took the data accordingly.
Experiment 2
- We started with the ultra violet 1st order and went down the spectrum collecting the stopping potentials for the different colors in the spectrum with no filter except on the green and yellow which we added the green and yellow filters respectively, only taking 1 reading this first time.
- We then moved on to the 2nd order color spectrum. We had to refocus the light after we started to take the 2nd order due to the fact that it would change the length between the light and the detector. We started again with the ultra violet and moved on down the line adding filters to the green and yellow respectfully. We only took one data reading this time. We had to adjust the focus when we went from blue to green.
Data
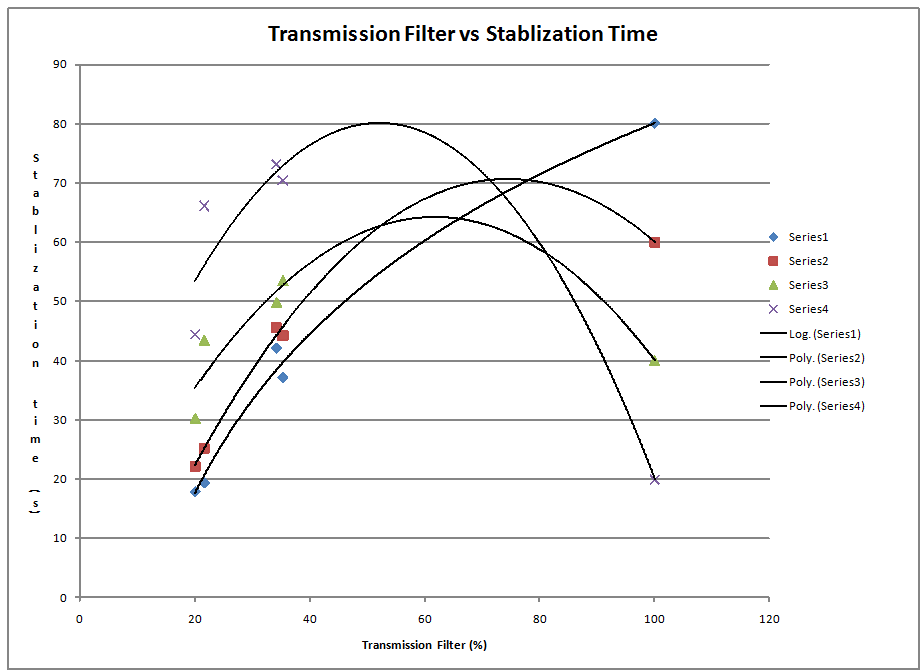
I took the following chart from Alexandra Andrego to show the wave lengths and frequency's of the emitted light
Color Frequency (Hz) Wavelength (nm) Yellow 5.18672E+14 578 Green 5.48996E+14 546.074 Blue 6.87858E+14 435.835 UV 1(or Violet) 7.40858E+14 404.656
SJK 22:32, 22 October 2009 (EDT)

I cannot tell where this graph came from? For one thing the axes must be mislabeled? The data you have in your google spreadsheet look much better than this...?
I used excel to do this graph and series 1 is violet, series 2 is blue, series 3 is green, series 4 is yellow. I used different trend line functions to fit the data for the different functions.
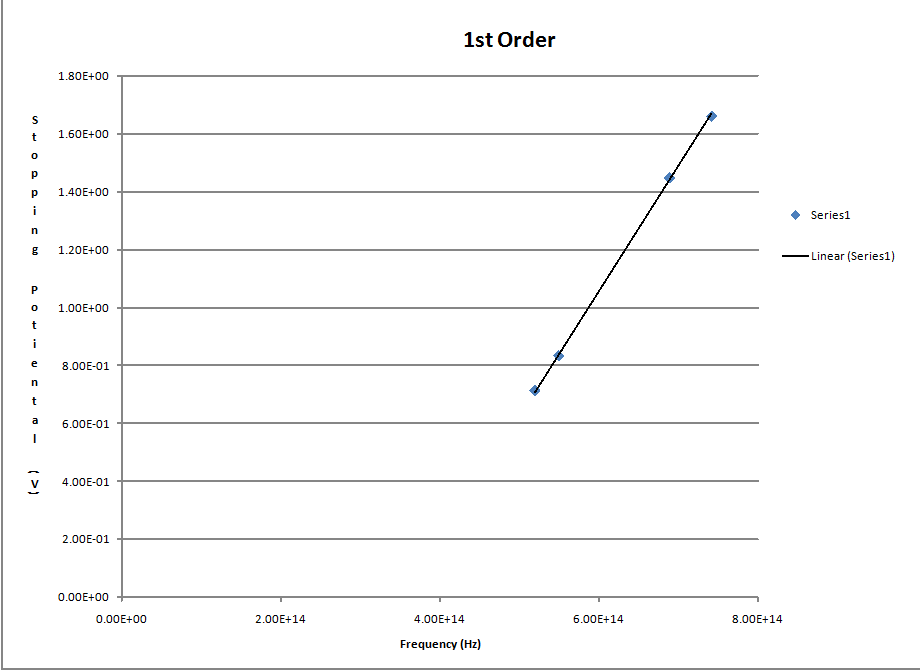 I used Excel to create this graph and used the trend line function to fit the line witch was the linear fit becouse it fit the data the best
I used Excel to create this graph and used the trend line function to fit the line witch was the linear fit becouse it fit the data the best
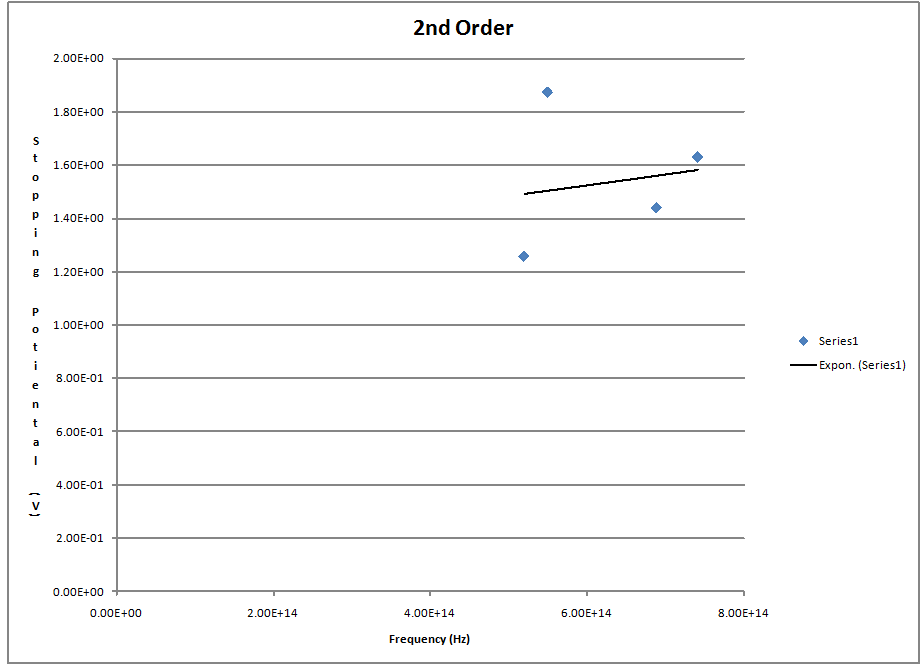 I again used excel to plot the data and fit the trend line again but this time i could not get as good of a fit and i used an exponential fit which did not match the data very well
I again used excel to plot the data and fit the trend line again but this time i could not get as good of a fit and i used an exponential fit which did not match the data very well
Finding a value for h/e
I am borrowing this way to find the values of h/e from Alexandra Andrago
- The total maximum energy of the electrons leaving the cathode in our experiment can be calculated using the following:
- [math]\displaystyle{ E =h \nu= KE_{max} + W_0 \,\! }[/math]
- [math]\displaystyle{ KE_{max}=\frac{1}{2}m_ev^2 }[/math]
- where [math]\displaystyle{ E=h\nu\,\! }[/math] is the initial energy of the photon
- and [math]\displaystyle{ E=KE_{max}+W_0\,\! }[/math] is the resulting energy (the final kinetic energy of the electron plus the energy loss due to the overcoming the work function)
- [math]\displaystyle{ m_e\,\! }[/math] is the rest mass of the electron and [math]\displaystyle{ v\,\! }[/math] is the final velocity of the electron
- The negative potential, [math]\displaystyle{ V_s\,\! }[/math], needed to stop the flow of electrons is derived by equating the potential barrier, [math]\displaystyle{ :eV_s\,\! }[/math], to the electron's kinetic energy where [math]\displaystyle{ e\,\! }[/math] is the charge of an electron and
- [math]\displaystyle{ eV_s=KE_{max}\,\! }[/math]
- So...
- [math]\displaystyle{ E=eV_s+W_0=h\nu\,\! }[/math]
- [math]\displaystyle{ eV_s=h\nu-W_0\,\! }[/math]
- [math]\displaystyle{ V_s=\frac{h\nu-W_0}{e}\,\! }[/math]
- From this we can see that there is a linear relation between the stopping potential [math]\displaystyle{ V_s\,\! }[/math] and the frequency [math]\displaystyle{ \nu\,\! }[/math]
- We can find the slope of this equation to be [math]\displaystyle{ \frac{h}{e}\,\! }[/math].
- Using the slope from our best-fit line and the electron's charge, [math]\displaystyle{ e\,\! }[/math], we can approximate the value of Planck's constant to be
- [math]\displaystyle{ e=1.602\times {10^{-19}} C\,\! }[/math]
- [math]\displaystyle{ h=me\,\! }[/math]
- where [math]\displaystyle{ m\,\! }[/math] is the slope of our line.
- So...
SJK 22:43, 22 October 2009 (EDT)

This has to be a mistake? I fit your data (which looked very nice for 1st order) and got 4.49 +/- 0.04 for the slope...which is much closer to the accepted value...
- [math]\displaystyle{ m_{first order}=2.3 \times 10^{-15} Vs\,\! }[/math]
- [math]\displaystyle{ h_{measured, first order}=me=(2.3\times 10^{-15} Vs)(1.602\times {10^{-19}} C)\,\! }[/math]
- [math]\displaystyle{ \simeq 3.68\times 10^{-34} Js\,\! }[/math]
- [math]\displaystyle{ m_{second order}=5.22\times 10^{-15} Vs\,\! }[/math]
- [math]\displaystyle{ h_{measured, second order}=me=(5.22\times 10^{-15} Vs)(1.602\times {10^{-19}} C)\,\! }[/math]
- [math]\displaystyle{ \simeq 8.36\times 10^{-34} Js\,\! }[/math]
- By using the y-intercept from our graph we can find the work function [math]\displaystyle{ W_0\,\! }[/math] for our equation
- [math]\displaystyle{ y=mx+b\,\! }[/math]
- [math]\displaystyle{ y_{intercept}=\frac{W_0}{e}\,\! }[/math]
- [math]\displaystyle{ W_0=ey_{intercept}\,\! }[/math]
- [math]\displaystyle{ y_{first order}=(2.3\times 10^{-15})x-3.5\,\! }[/math]
- [math]\displaystyle{ y_{intercept, first order}=-3.5\,\! }[/math]
- [math]\displaystyle{ W_{0measured, first order}=(-3.5 V)(1.602\times {10^{-19}} C)\,\! }[/math]
- [math]\displaystyle{ \simeq -5.607\times 10^{-19} J\,\! }[/math]
- [math]\displaystyle{ y_{second order}=(5.22\pm 0.001\times 10^{-15})x-0.97\pm 0.001\,\! }[/math]
- [math]\displaystyle{ y_{intercept, second order}=-0.97\pm 0.001\,\! }[/math]
- [math]\displaystyle{ W_{0measured, second order}=(-0.97\pm 0.001 V)(1.602\times {10^{-19}} C)\,\! }[/math]
- [math]\displaystyle{ \simeq -1.55\times 10^{-19} J\,\! }[/math]
SJK 17:25, 25 October 2009 (EDT)

In addition to it looking like you made a mistake (I get a different fit result than you list), you don't seem to have looked at the results very carefully. For example, I don't see any discussion of 2nd order versus 1st order. Do you expect them to be different? Does it look like there's a big problem with the 2nd order green? Does it make any sense at all to average to very different slopes and report that answer?
Recalculations
- I went back and recalculated the values for the slope and this is what I got
- 1st order [math]\displaystyle{ m*e=4.31(.046)\times 10^{-15}V }[/math]
- and the value for [math]\displaystyle{ me=(4.31(.046)\times 10^{-15}V s)(1.602\times 10^{-19}C)\simeq 6.904(.074)Js }[/math]
- 2nd order [math]\displaystyle{ m*e=2.974(.709)\times 10^{-15}Vs }[/math]
- and thus for the value [math]\displaystyle{ me=(2.974(.709)\times 10^{-15}V s)(1.602\times 10^{-19}C)\simeq 4.765(1.135)Js }[/math]
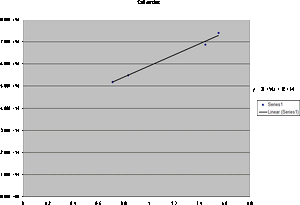
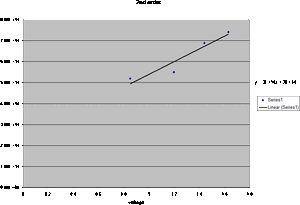
- From the new graphs we find that the y-intercept is 2*10^14 for the 2nd order and 3*10^14. Taking this and multiplying by the value of e we obtain 4.806*10^-5 for the first order and 3.204*10^-5 for the second order. Taking this and multiplying it by the value for e again we obtain
7.7*10-24J for the first order and 5.13*10^-24J for the second order.
Errors
- one possible source for error is that from touching the filters and getting fingerprints or dirt/grime on them. this would prevent some light form getting to the detector and interfere with the readings and the time it takes to charge the capacitor.