BIO254:Gprotein: Difference between revisions
| Line 43: | Line 43: | ||
==Small G Proteins== | ==Small G Proteins== | ||
In contrast to heterotrimeric G proteins, small G proteins are monomeric. Small G proteins are between 25 and 40 kD, which is indeed smaller than the heterotrimeric G proteins, the alpha subunit of which alone is 45 kD. There are over 100 small G proteins. The small G protein superfamily includes the Ras family (signal transduction), the Rho/Rac family (cytoskeleton), the Rab and Sar1/Arf families (vescicle trafficking), and the Ran family (nuclear import/export) (Takai <i>et al.</i>, 2001). | In contrast to heterotrimeric G proteins, small G proteins are monomeric. Small G proteins are between 25 and 40 kD, which is indeed smaller than the heterotrimeric G proteins, the alpha subunit of which alone is 45 kD. There are over 100 small G proteins. The small G protein superfamily includes the Ras family (signal transduction), the Rho/Rac family (cytoskeleton), the Rab and Sar1/Arf families (vescicle trafficking), and the Ran family (nuclear import/export) (Takai <i>et al.</i>, 2001). Like heterotrimeric G proteins, small G protein function involves activation by binding to GTP and release of GDP and inactivation by hydrolyis of GTP to GDP. The kinetics of these steps are such that small G proteins can and do act as biological timers. The mechanism of small G protein function is demonstrated in Figure 5 for the case of the Rho protein. [[Image:Luo_rho_schem.jpg|thumb|300px|center|Figure 5. Image taken from Luo, 2000.]] | ||
The mechanism of small G | |||
====The Rho/Rac family of small GTPases==== | ====The Rho/Rac family of small GTPases==== | ||
Revision as of 10:45, 27 October 2006
Introduction
The term G protein refers to proteins that bind the nucleotide guanine as guanosine triphosphate (GTP) and guanosine diphosphate (GDP). There are two types of G proteins: heterotrimeric, or large, G proteins and small G proteins. Heterotrimeric G proteins are membrane-associated and, along with G protein-coupled receptors (GPCRs), function primarily in cell signalling and signal transduction. Small GTP-binding proteins function in diverse cellular processes including signal transduction, cytoskeletal reorganization, and vesicle trafficking (Takai et al., 2001).
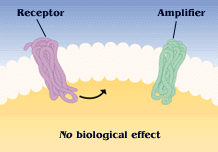
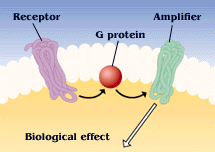
A molecular switch
G protein activity is dependent on whether it is binding GTP or GDP. This useful property has led to the appropriation of G proteins by many cellular processes to be used as "molecular switches". G proteins are generally thought to be "active" when binding GTP and "inactive" when binding GDP. The transition from the GTP-bound state to the GDP-bound state depends on the hydrolysis of GTP. This GTPase activity is either completely intrinsic to the G protein or is enhanced by another class of proteins, "GTPase activating proteins" (GAPs). The GDP to GTP transition requires the dissociation of GDP, so that GTP may again bind at the active site. Proteins that mediate this GDP dissociation are known as guanine nucleotide exchange factors (GEFs).
Heterotrimeric G proteins
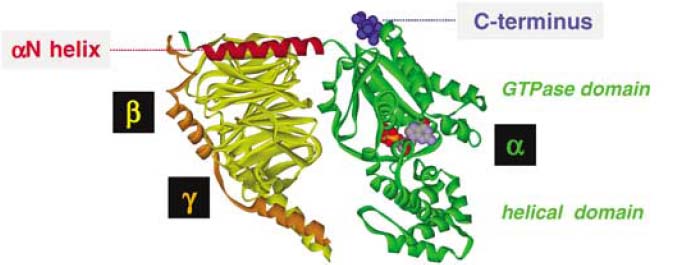
Discovery
The heterotrimeric G protein that Gilman isolated increased cAMP levels. In 1980 Martin Rodbell wrote a review (Rodbell, 1980) that helped direct the search for the first cAMP reducing G-protein to be discovered, in 1984. Martin Rodbell and Alfred G. Gilman were awarded the 1994 Nobel Prize in Physiology and Medicine for the discovery of "G-proteins and the role of these proteins in signal transduction in cells". Since the first G-proteins were identified, many others with effectors other than cAMP have been cloned, in many cases by homology. Currently 16 alpha, 5 beta, and 14 gamma subunits have been identified (Milligan, 2006).
G Protein-Coupled Receptors
Heterotrimeric G proteins associate with 7-transmembrane domain receptors called G protein-coupled receptors (GPCRs) at the cell membrane. There are as many as 865 GPCR-encoding genes in humans (Milligan, 2006). Specific GPCRs are recognized by specific G proteins. This recognition is mediated by a sequence at the C-terminus of the G-protein α subunit. For more information on G protein-coupled receptors, see the GPCR wikipedia entry
Mechanism
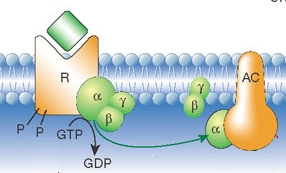
Families
Effectors
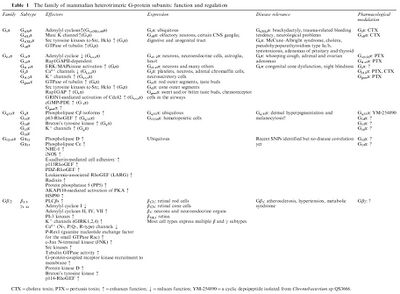
The original GPCR cell signaling pathway described was a Gs protein that activates adenylate cyclase. Certain Gi pathways are characterized by the ability of Gαi to inhibit adenylate cyclase. Gβγ subunits have their own downstream effectors, which include phosphatidylinositol 3-kinase (PI3K). Certain Gq pathways act through inositol trisphosphate (IP3), diacylglycerol (DAG), and protein kinase C (PKC). The Gα12 and Gα13 family effectors include phospholipases.
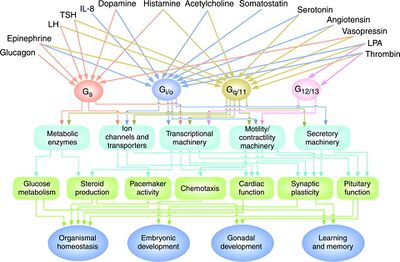
Roles in the Nervous System
Many heterotrimeric G proteins are specific to certain cell types and tissues (Table 1). Certain heterotrimeric G proteins are expressed specifically in nervous system components including olfactory neurons, CNS ganglia, neuroendocrine cells, astroglia, and retinal rod and cone cells. In the nervous system heterotrimeric G proteins are found in signaling pathways mediated by dopamine, epinephrine, serotonin, glucagon, light, olfactory signals, and other factors. They are involved in taste, vision, affect, arousal, and other functions.
Small G Proteins
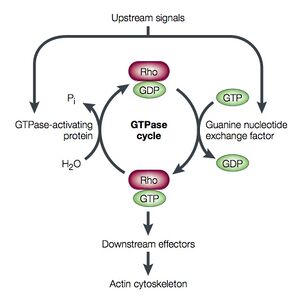
The Rho/Rac family of small GTPases
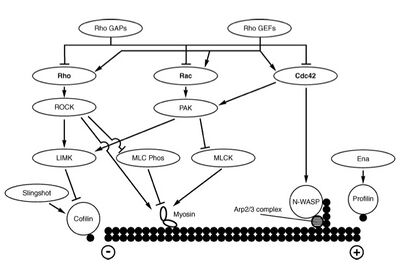
References
1. Takai Y, Sasaki T, Matozaki T. Small GTP-Binding Proteins. Physiol Rev. 81, 153-208 (2001).
2. Milligan G, Kostenis E. Heterotrimeric G-proteins: a short history. Br J Pharmacol. 147 Suppl 1:S46-55 (2006)
3. Northup JK, Sternweis PC, Smigel MD, Schleifer LS, Ross EM, Gilman AG. Purification of the regulatory component of adenylate cyclase. Proc. Natl. Acad. Sci. U.S.A. 77 6516–6520 (1980).
4. Rodbell, M. The role of hormone receptors and GTP regulatory proteins in membrane transduction. Nature 284, 17–22 (1980).
5. Firestein, S. How the olfactory system makes sense of scents. Nature 413, 211-218 (2001)
6. Neves S, Ram P, Iyengar R. G protein pathways. Science 296, 1636-1639 (2002)
7. Luo L. Rho GTPases in neuronal morphogenesis Nat Rev Neurosci. 1, 173-180 (2000).
8. Huber A, Kolodkin A, Ginty D, Cloutier JF. Signaling at the growth cone: ligand-receptor complexes and the control of axon growth and guidance. Ann Rv Neurosci 26, 509-63 (2003)
External Links
Recent updates to the site:
List of abbreviations:
- N
- This edit created a new page (also see list of new pages)
- m
- This is a minor edit
- b
- This edit was performed by a bot
- (±123)
- The page size changed by this number of bytes
2 April 2026
|
|
19:57 | (Upload log) [Xinyu Liu; David Altman (8×)] | |||
|
|
19:57 David Altman talk contribs uploaded File:LabPicSpring2026.jpeg | ||||
|
|
19:57 David Altman talk contribs uploaded File:OASposterSpring2026.jpeg | ||||
|
|
19:56 David Altman talk contribs uploaded File:OAStalkSpring2026.jpeg | ||||
|
|
19:42 David Altman talk contribs uploaded File:Soto WUbites Spring 2026.pdf | ||||
|
|
19:42 David Altman talk contribs uploaded File:Shipp WUbites Spring 2026.pdf | ||||
|
|
19:42 David Altman talk contribs uploaded File:Margerum WUbites Spring 2026.pdf | ||||
|
|
19:42 David Altman talk contribs uploaded File:Bentley WUbites Spring 2026.pdf | ||||
|
|
19:40 David Altman talk contribs uploaded File:Bashioum WUbites Spring 2026.pdf | ||||
|
|
09:27 Xinyu Liu talk contribs uploaded File:Small2026.jpg | ||||
| 19:56 | Altman:Pictures diffhist +354 David Altman talk contribs | ||||
| 19:50 | Altman diffhist −54 David Altman talk contribs (→News) | ||||
|
|
19:40 | Altman:WUbites 2 changes history +992 [David Altman (2×)] | |||
|
|
19:40 (cur | prev) +40 David Altman talk contribs (→WUbites) | ||||
|
|
19:39 (cur | prev) +952 David Altman talk contribs (→WUbites) | ||||
| 18:36 | CHIP:Talks diffhist −19 Gabor Balazsi talk contribs | ||||
|
|
09:28 | Gao lab:Publications 2 changes history +549 [Xinyu Liu (2×)] | |||
|
|
09:28 (cur | prev) +24 Xinyu Liu talk contribs (→Publications) | ||||
|
|
09:26 (cur | prev) +525 Xinyu Liu talk contribs (→Publications) | ||||
1 April 2026
|
|
11:32 | Hu 2 changes history +121 [Hugangqing (2×)] | |||
|
|
11:32 (cur | prev) +2 Hugangqing talk contribs | ||||
|
|
11:32 (cur | prev) +119 Hugangqing talk contribs | ||||