User:Allison K. Alix/Notebook/Thesis Research/2013/04/09: Difference between revisions
(→Data) |
(→Data) |
||
| Line 54: | Line 54: | ||
Part 2: | Part 2: | ||
[[Image:ThiolDNA_MB_pic_UV-Vis.png]] | |||
UV-Vis Spectrum of thiol-DNA/MB hybrid | |||
[[Image:ThiolDNA_MB_pic.png]] | [[Image:ThiolDNA_MB_pic.png]] | ||
| Line 60: | Line 64: | ||
Part 3: | Part 3: | ||
[[Image:ThiolDNA_ThT_pic_UV-Vis.png]] | |||
UV-Vis Spectrum of thiol-DNA/ThT hybrid | |||
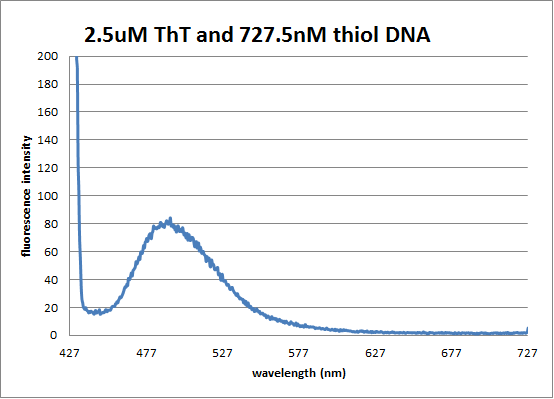
[[Image:ThiolDNA ThT pic.png]] | [[Image:ThiolDNA ThT pic.png]] | ||
Fluorescence Spectrum of thiol-DNA/ThT hybrid | Fluorescence Spectrum of thiol-DNA/ThT hybrid | ||
Revision as of 07:39, 12 April 2013
| <html><img src="/images/9/94/Report.png" border="0" /></html> Main project page <html><img src="/images/c/c3/Resultset_previous.png" border="0" /></html>Previous entry<html> </html>Next entry<html><img src="/images/5/5c/Resultset_next.png" border="0" /></html> | |
Objectives
ProceduresPart 1: Preparation of 1mL of 2.98μM ethane thiol WARNING: ethanethiol is very volatile. Handle under the hood only wearing PPE.
1) Find the concentration of 0.0125mL pure ethanethiol in 5mL water 0.839g/mL x 0.0125mL = 0.0104875g 0.0104875g/(62.13 g/mol) = 1.68799 x 10-4 mol 1.68799 x 10-4 mol/5mL x (1000mL/1L) = 0.034M = 34mM
340mM (x) = (30mM)(1mL) x = 0.088mL = 88μL in 912μL H2O 30mM (x) = 3mM (1mL) x = 100μL in 900μL 3mM(x) = 298μM (1mL) x = 99μL in 901μL to obtain 298μM ethanethiol 2) Reacting AuNP with ethanethiol
Part 2: Measure absorbance/fluorescence of AuNP-MB Part 3: Measure absorbance/fluorescence of AuNP-ThT DataPart 2: UV-Vis Spectrum of thiol-DNA/MB hybrid Fluorescence Spectrum of thiol-DNA/MB hybrid Part 3: UV-Vis Spectrum of thiol-DNA/ThT hybrid Fluorescence Spectrum of thiol-DNA/ThT hybrid
| |