IGEM:Harvard/2006/Cyanobacteria/Notebook/2006-8-12
<html><style type='text/css'> .tabs {
font-size:80%;
font-weight:none;
width: 100%;
color: #FFFFFF;
background:#FFFFFF url("/images/5/54/DarkgreenTab-bg.gif") repeat-x bottom;
}
.tabs li {
background:url("/images/3/36/DarkgeenTab-left.gif") no-repeat left top;
}
.tabs a,.tabs strong {
background:url("/images/d/d3/DarkgreenTab-right.gif") no-repeat right top;
color:#FFFFFF;
padding: 3px 10px 3px 4px;
}
.tabs strong{
color:#CCFF00;
background-image:url("/images/b/b1/DarkgreenTab-right_on.gif");
}
.tabs a:hover{
color:#66FF00;
}
</style></html>
To-do
RFP in low-copy plasmid (transformed in Top10 and Top10F')- GFP dev in low-copy plasmid (from digest assay)
Run the redigest of pSB4A3 (pSB4A3\S-P, pSB4A3\X-P, psB4A3 undigested) on a gel, to see if the plasmid is the right length or if it has an insertOur gel boiled in the tank; we've labeled the tank armed and dangerous. We lost this DNA.CIP treat the 4 gel-extracted pSB4A3\S-P from the digest assay- If the pSB4A3 plasmid looks alright, ligate GFP dev\X-P + psB4A3\S-P
- Transform ligation products
- Kai\X-P + J04500\S-P ligation
Run the KaiA\X-P, KaiB\X-P, and KaiC\X-P digests from yesterday on a gelOur gel boiled in the tank; we've labeled the tank armed and dangerous. We lost this DNA.Redo the KaiA\X-P, KaiB\X-P, and KaiC\X-P digests- Run the KaiA\X-P, KaiB\X-P, and KaiC\X-P digests on a gel
- Extract and purify KaiA\X-P, KaiB\X-P, and KaiC\X-P
CIP treat J04500\S-P- Ligate Kai\X-P + J04500\S-P
- Transform ligation products
Growing up Kai--Zsun 17:37, 12 August 2006 (EDT)Inoculate cultures of the transformed KaiA/B/C GeneART plasmids
- More backbone
- Grow up a lot of pSB4A3- and J04500- transformed cells for midiprepping
Results of Restreak of J00450+PSB4A3
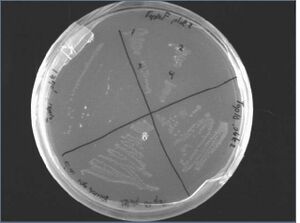
Although the colonies took two days to grow, the restreak of the plates on Carb worked; we will now verify the ligation via PCR while growing liquid culture from them.
PCR ligation test of J04450+PSB4A3 + innoculation
Following Perry's Protocol which worked pretty well, in order to check the ligation we will do a PCR using the PCR Supermix from the VF2 and VR areas of the plasmid;
From the above image, we will try 7 colonies:
- Top10 plate2 A (eg. 102)
- Top10 plate1 A (eg. 101)
- Top10F plate1 (eg. F1)
- Top10F plate2 1 (eg. F2-1)
- Top10F plate2 2 (eg. F2-2)
- Top10F plate2 3 (eg. F2-3)
- Top10F plate2 3 (eg. F2-4)
From Perry's Protocol, each will have:
- 8uL PCR supermix
- 1uL VF2 (2uM)
- 1uL VR (2uM)
Then, get a carb plate, pick the desired colony, grow in 8mL LB+Carb. Choose another (monoclonal source), dip it into the 10uL rxn.
The runtime protocol, modified from Perry's is:
- 95@15m
- Loopx30
- 95@30
- 55@30
- 72@1.30
- 72@10
- 4@forever

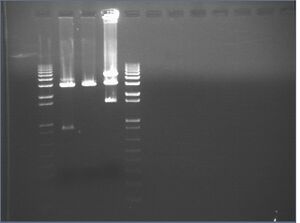
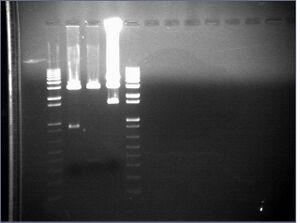
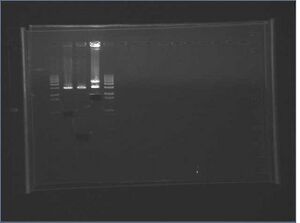
Redigest of Kai\X-P
Since our gel failed and we lost all of our digest product, we redigested KaiA/B/C with X-P, following the same reactions as yesterday.
- 4 uL DNA (~800 µg DNA)
- 17.25 uL H2O
- 2.5 uL buffer (Buffer 3)
- 0.5 uL enzyme 1 (XbaI)
- 0.5 uL enzyme 2 (PstI)
- 0.25 uL BSA
The digest ran for 2 hours at 40C.
CIP treatment of pSB4A3\S-P, J04500\S-P, and pSB1A2\X-P
Yesterday we gel-purified three digested backbones: pSB4A3\S-P, J00450\S-P, and pSB1A2\X-P. Normally one does the CIP treatment immediately after the digest instead of gel-purifying first. Since we didn't add CIP then, we had to add it now.
We didn't want to do another gel purification after this step because our yield would likely be very low (the backbones have around 3 ng/µL concentration of DNA). Therefore we had to inactivate the CIP by heat.
- Add 0.1 µL CIP to each digest product
- Incubate at 37C for 1 h
- Add EDTA to 5 mM
- Heat shock at 75C for 15 min
KaiA/B/C\X-P digest gel