Talk:20.109(S09):Induce protein and evaluate DNA (Day5)
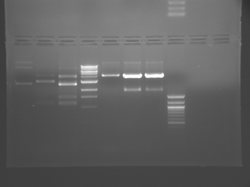
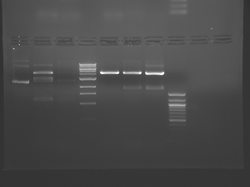
Improved Fsp Digest
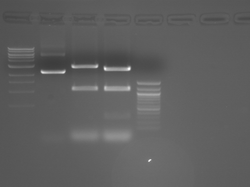
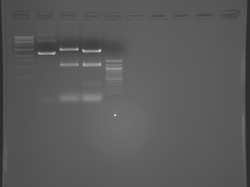
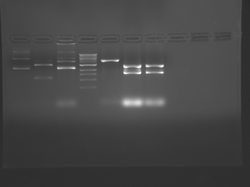
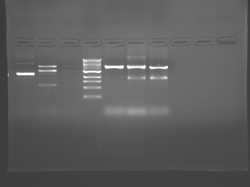
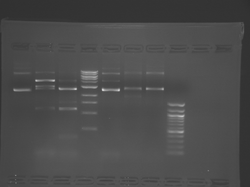
Lane 1 is the 1 Kbp ladder, lane 5 the 100 bp ladder.
Lanes 2, 3, and 4 are uncut IPC (WT), digested IPC, and digested IPC-S101L, respectively.
Samples were digested overnight, as FspI appears to digest somewhat weakly otherwise.
TR O/N cultures and Gels
The table below lists the OD600 values of 1:10 dilutions of the cultures grown overnight, as well as the pellet observations.
| Group | OD WT | OD S101L | OD Chosen mutant | Pellets yellow? |
|---|---|---|---|---|
| Pink | 0.117 | 0.098 | 0.126 | Yes |
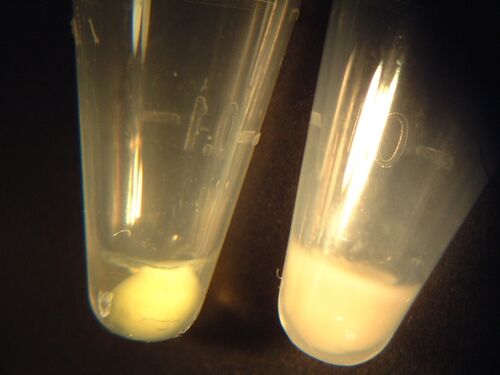
| Yellow | 0.118 | 0.091 | 0.148 | WT and S101L bright yellow-green; Y64D paler matte yellow |
| Red | 0.121 | 0.187 | 0.129 | Yes |
| Orange | 0.117 | 0.087 | 0.122 | WT and S101L bright yellow-green; E31I paler matte yellow |
| Purple | 0.109 | 0.075 | 0.121 | Yes |
| Green | 0.106 | 0.095 | 0.072 | Yes |
| Grey | 0.111 | 0.102 | 0.148 | All bright yellow-green |
Note: 1% gels were run for 45 min, 1.2% gels for 60 min, both at 100V.
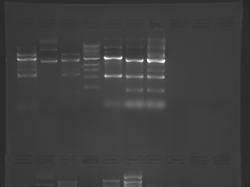
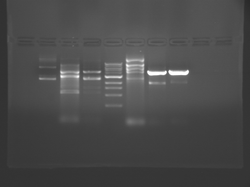
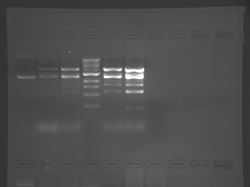
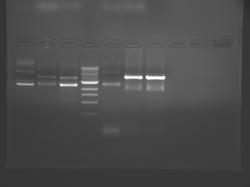
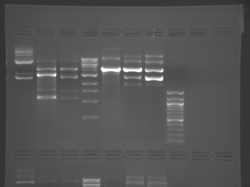
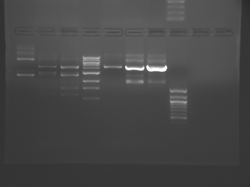
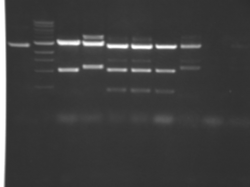
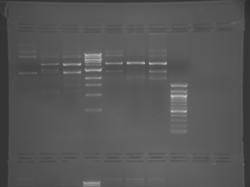
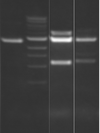
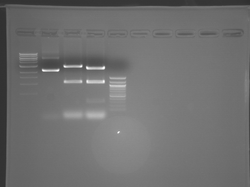
There seems to have been a contamination or other strange issue with the S101L DNA and/or FspI enzyme. For your final reports, please use my old data, which is posted above. Unfortunately, some of the bands are faint (this experiment pre-dates our modern gel doc system). Lane 1 is parent IPC (only one form of the plasmid is visible), lane 2 is the ladder, lane 4 is the parent digested with FspI, and lane 8 is the S101L mutant digested with FspI. The short band is very faint, but it is present. I will have the original photo available for viewing for anyone who wants to clearly see the short band. At right is a cropped version for easier viewing. Update: Upon reflection, most of the S101L data does not look nearly as bad as I thought. As you interpret your data, keep in mind what are all the possible band sizes you could get. What if the enzyme only partially digests? Nevertheless, I will try to re-run this experiment to get a better photo for your reports in the coming days.
WF O/N cultures and Gels
The table below lists the OD600 values of 1:10 dilutions of the cultures grown overnight, as well as the pellet observations.
| Group | OD WT | OD S101L | OD Chosen mutant | Pellets yellow? |
|---|---|---|---|---|
| Pink | 0.105 | 0.098 | 0.105 | Yes |
| Purple | 0.244 | 0.121 | 0.122 | Yes |
| Red (O/N) | 0.229 | 0.284 | 0.282 | WT and S101L bright yellow-green, mutant white |
| Grey (O/N) | 0.206 | 0.106 | 0.109 | All are bright yellow-green |
| Green | 0.245 | 0.099 | 0.115 | all are yellow/green |
| Yellow | 0.238 | 0.120 | 0.116 | all are bright yellow-green |
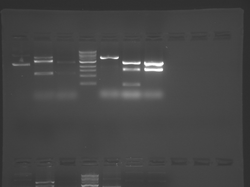
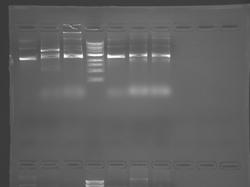
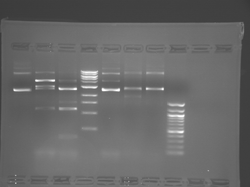
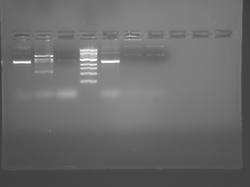
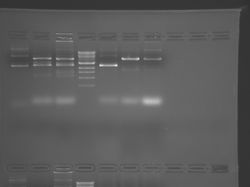
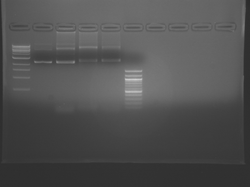
Please see notes about S101L in T/R section above.