Todd:Chem3x11 ToddL12
Chem3x11 Lecture 12
This lecture introduces pericyclic reactions and specifically electrocyclic reactions.
(Back to the main teaching page)
Key concepts
- Pericyclic reactions involve cyclic transition states
- They proceed with no intermediates, and are stereospecific
- Electrocyclic reactions involve a π system giving a ring (extra σ) with one less π bond
Some Opening Experimental Observations
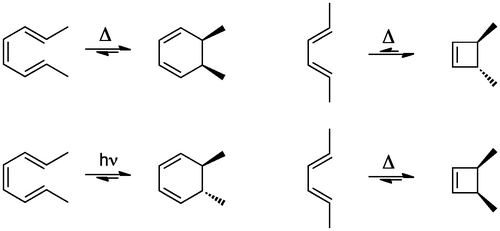
Here are some reactions we've not seen before. The outcomes are, on the face of it, difficult to understand, particularly the stereochemical outcomes which are very specific. There are no apparent intermediates. Solvent polarity has little influence on the outcome. The reactions are equilibria. Imagine you were presented with these reactions and asked to explain the mechanism.
This was the situation in the 1960's when an explanation for these reactions, and several other types, finally emerged. The key was to look at the orbitals involved (an approach taken by Woodward and Hoffmann) and this was later simplified just to a consideration of the Frontier Orbitals (i.e., the HOMO and LUMO) (an approach pioneered by Fukui). To understand this, let's make sure we can draw the relevant frontier orbitals for molecules like this.
(Note if you read further about pericyclic reactions you'll see three competing models for understanding them - a) Orbital correlation, b) Frontier Molecular Orbital (FMO) Theory and c) Aromatic Transition State Analysis. Typically FMO is what we'll be dealing with, because of its economy and simplicity.)
Frontier Orbitals of Polyenes
The σ framework of an organic molecule contains the strong (low energy) bonds, meaning the corresponding antibonding orbitals are high in energy. Our analysis of frontier orbitals in reactions of this kind typically focusses on the π system, where the higher energy bonding interactions are, as well as the lowest-lying antibonding molcular orbitals. If we therefore forget about the σ bonds and just focus on the π bonds, we can easily sketch the HOMO and LUMO for ethene.
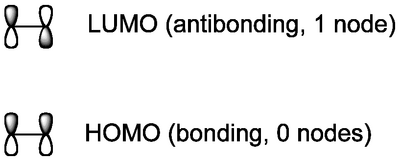
(Remember that the MO's will actually look like clouds spread over the whole molecule, rather than just separate atomic AOs, but there will be nodes for every MO above the HOMO, as we'll see, where the constituent AO's are not in phase - as for the LUMO above)
In the case of butadiene, there are four π molecular orbitals, two bonding and two antibonding. The way to evaluate how they are drawn is to generate an increasing number of nodes.
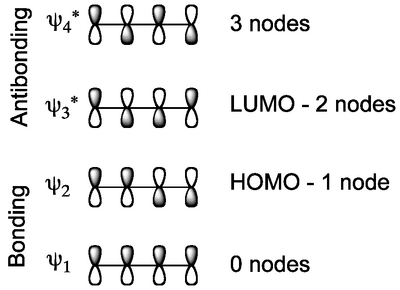
Based on the number of electrons in the π system, the HOMO and LUMO must be as shown. In the ground state of the molecule, the electrons will just populate the bonding molecular orbitals, as expected. For hexatriene, the analysis is just the same:

So being able to sketch all of these means that we know we have identified the sign of the HOMO and LUMO on each carbon. This will be important for reactivity. Now let's go back and look at one of the reactions in Scheme 1 in a little more detail.
How the Frontier Orbitals Guide an Example Reaction
The reaction we'll start with is the transformation of that octatriene to the cyclohexadiene shown in Scheme 1. The starting material is a triene with methyl groups on the end. The mechanism is just a cyclic rearrangement of electrons, resulting in one less π bond and one more σ bond (largely explaining the position of equilibrium). This reaction type is called an Electrocyclic Reaction and is one of the reactions we'll consider. The transition state has a cyclic arrangement of electrons, like an electric circuit. We're going to see various types of these reactions with small variations, but if they involve a cyclic arrangement of electrons in the transition state we call them pericyclic reactions generally.

This simple 2D representation does not explain what's really going on, nor the stereochemical outcome. Notice that there is a σ bond being formed from two π bonds. Since the electrons that are moving around are in the HOMO, we just need to consider that frontier orbital. A way to think about what's going on is that the π orbitals at the ends of the triene are "rotating" so they overlap and form a new σ bond, thereby creating the ring. If we look back at our HOMO diagram for the triene (Scheme 4) we see that these end lobes have the same sign. If they are to productively overlap to give a new σ bond, lobes of the same sign (symmetry) must come together. This specifies a direction of rotation for this reaction to work, in this case the direction of rotation is the opposite way for each lobe, and we call this disrotatory. The need for a specific rotation in the bonds mandates that the two methyl groups end up on the same face of the ring.

So in this case the reaction shown is possible because the interacting MO lobes are of the correct sign. We say the reaction is symmetry allowed. Let's test this idea with the second reaction in Scheme 1, generating the 4-membered ring. Looking back to our MO diagram, we find that the end lobes are of opposite sign, or orientation, meaning that for a productive bond forming interaction there needs to be rotation of the C-C bonds in the same direction, and we call this conrotatory. Again, this is a stereospecific outcome - the stereochemistry is set by an orbital requirement.

Ring-Opening Reactions Obey the Same Rules
The reactions we're considering are reversible - empirically we see them going in both directions. Based on the principle of microscopic reversibility the mechanisms must involve the same kinds of orbital interactions. So we ought to expect the corresponding ring opening reactions to be understandable using a similar analysis to the above. That's the case. As an example let's consider the opening of the cyclobutene that was the product of the reaction in Scheme 1. Here there is not one extended π MO, but instead a σ bond and a π bond. Let's look at how the LUMO of the π bond can interact with the HOMO of the σ bond. (Do you get the same result if you consider the orbitals the other way round?). For a bonding interaction, the same conrotatory motion needs to be seen.

You may ask yourself why the rotation happens in one direction rather than the other (given that we could just imagine the LUMO the other way up) and the answer is beyond the scope of this course (many cases are governed by sterics, but there are some neat electronic cases that give odd results). However, based on what you now know, you might want to think about why the bicyclic molecule Dewar benzene, shown, doesn't open to give benzene, despite being so obviously strained.
Photochemical Version of Electrocyclic Reactions
The reactions above were driven by heat. We ought to call them thermal electrocyclic reactions. It's also possible to make these reactions go by shining light on the reagents. If you do that, you see that the stereochemical outcome is altered, as shown in Scheme 1.
Now many reactions that are driven by light (photochemical reactions) do not obey the same kinds of rules as their thermal counterparts because there are radical intermediates formed and these take part in mechanisms that are not pericyclic, but are instead stepwise processes. However, there are some photochemical electrocyclic reactions, and there is a model for understanding the outcome.
The key is to realise that the reaction proceeds because an electron from the HOMO is promoted to the LUMO through the impact of the photons. This means the orbital that was the LUMO becomes the HOMO, and this then changes the symmetry of the interaction of the end lobes of the π system.

...and the same idea can be applied to the triene example - I'll leave that as an exercise.
One can extrapolate to a set of rules if desired:
If there is an even number of π bonds in the open chain compound, thermal electrocyclic reactions go conrotatory. An odd number of π bonds goes disrotatory. The opposite applies for photochemical reactions.
However, it's important to know how to check that this rule is right by being able to draw the HOMO of any given case and identifying the MO lobe overlap to give the symmetry-allowed reaction.
Examples
Matt Tarnowski, former 3111 students, says: Check out the [2+2] photocycloaddition reported in figure 2 of this article covering the synthesis of Steviol reported by P S Baran et al. in 2013.
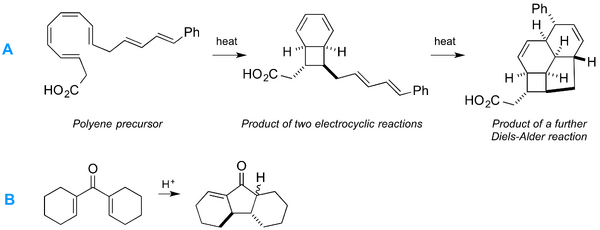
Below are two more examples to consider. The first (A) is the striking sequence of reactions involved in the synthesis of the endrianic acids. The sequence shown involves two electrocyclic reactions and one Diels Alder reaction (see Lecture 13). The stereochemistry is tightly controlled throughout, but not by any enzymatic catalysis (the compounds are naturally produced as racemates) but by the inherent orbital interactions involved. You can read a very nice summary of this chemistry in Clayden, chapter 35, or you can read the original proposal for this chemistry by David Black, and the experimental execution of it in the lab. The second, simpler, reaction (B) is known as the Nazarov cyclisation. Can you see how this can work via an electrocyclic mechanism? (you need to use the acid).
The Licence for This Page
Is CC-BY-3.0 meaning you can use whatever you want, provided you cite me.
