IGEM:IMPERIAL/2007/Experimental Design/Phase1/Results 2.1: Difference between revisions
| Line 62: | Line 62: | ||
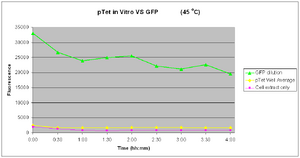
| width="200px"|[[Image:IC2007 Experimental Design PTet vitro 45degrees-4hours.PNG|thumb|300px|<font color=blue>22-08-2007</font>- Results of pTet at 45°C]] | | width="200px"|[[Image:IC2007 Experimental Design PTet vitro 45degrees-4hours.PNG|thumb|300px|<font color=blue>22-08-2007</font>- Results of pTet at 45°C]] | ||
| width="50px"| | | width="50px"| | ||
|width="400px"|We tested pTet-GFP in vitro in commcercial S30 extract at 45°C for 4 hours, sampleing every 30minutes. The results to the left show the followig | |width="400px"|We tested pTet-GFP in vitro in commcercial S30 extract at 45°C for 4 hours, sampleing every 30minutes. | ||
The results show that there minimal expression of GFP at 45°C. The sample only slightly increases above the negative control showing minimal expression. The positive control again shows expotential decay. Interestingly there is even greater variation within the results, giving very unpreditable trend lines. | |||
The results to the left show the followig | |||
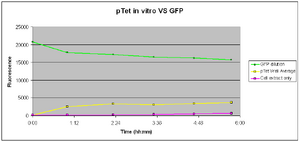
<div style="color:Green">Positive Control - Fluorescence of the diluted GFP solution</div> | <div style="color:Green">Positive Control - Fluorescence of the diluted GFP solution</div> | ||
<div style="color:#F88017"> The average fluorescence of the pTet samples(3)</div> | <div style="color:#F88017"> The average fluorescence of the pTet samples(3)</div> | ||
<div style="color:#C45AEC">Negative Control - The fluoresence of a solution containing only S30 cell extract</div> | <div style="color:#C45AEC">Negative Control - The fluoresence of a solution containing only S30 cell extract</div> | ||
|} | |} | ||
<br clear=all> | <br clear=all> | ||
Revision as of 08:37, 18 September 2007
In Vitro Tetsing
Aim
The aims of this experiment is to test the following constructs in vitro at 37°C To Determine if the following constructs work in vivo:
After this testing we will test working constructs at 10°C and 37°C. In addition we want to identify any problems with our testing method
Both constructs were tested in vitro on *pTet and pT7 in vitro Tested 21-08-2007 to Tested 23-08-2007
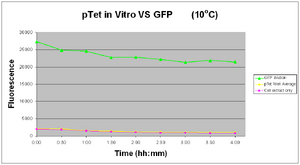
pTet was tested in vitro at 10°C and 45°C Tested 21-08-2007
Results
pTet-GFP(100ng/μl)
Test: 21-08-2007
37 Degrees
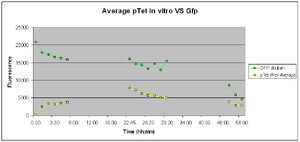
Test: 21-08-2007 to Test: 23-08-2007
37 Degrees
Test: 22-08-2007
10 Degrees
Test: 22-08-2007
45 Degrees
pT7-GFP In Vitro (100ng/μl)
37 Degrees
The pT7 was tested in vitro for a span of 4 hours at 37oC right after iPTG induction. After the initial reading, it was found that fluorescence decreased down to a steady level. This was observed for all our 3 samples and negative control, indicating that it was due to a change in the in vitro background fluorescence. The possible source of this decrease could be due to an extra experimental step taken, which was a quick centrifugation before the plate was read in the fluorometer.
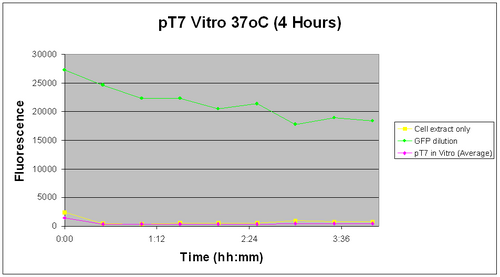
The graph on the right dispays the following.
As you can observe, the pT7 does not appear to be working in vitro either. The commercial S30 cell extract used does not promote it to start expressing GFP at least within the 4 hours during which our tests were carried out and its fluorescene levels remain well below the diluted GFP.
The plate containing the samples was stored in a 37oC incubator overnight. It was re-tested the next morning to see whether GFP had been expressed,22 hours after of induction. The results were joined with the initial testing done over the first 4 hours of induction and are shown below.
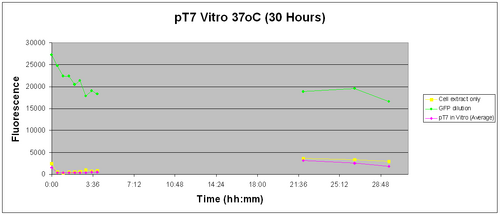
The graph legend is the same as the one of the graph above. The lab was closed between hours 4:00 and 22:00 and hence a large array of readings is missing. But we are only interested whether the construct works. With that in mind and given the slow degradation rate of GFP we should be able to detect if it was expressed even 20 hours later.
The fluorescence the next day (after 22 hours of induction) had risen a bit but so did our -ve control. This leads us to suspect that some of our samples had been contaminated perhaps with GFP from the +ve control. From this we realised we had to re-think the way our samples were arranged on the well plates. We had to allow more spacing betweeen the samples and avoid placing samples next to adjacent wells.
Overall though, the fluorescence readings were minimal compared to pTet.It can thus be concluded that the pT7 construct does not work in vitro with the commercial S30 cell extract.
| Complete set of results and raw data |