User:Andy Maloney/Surface passivation effects on kinesin and microtubules
Purpose
This page describes the experiment where I investigated gliding speed variations of microtubules using different surface passivation schemes using the gliding motility assay.
This is the second chapter in my completely open notebook science dissertation. If you would like to post questions, comments, or concerns, please join the wiki and post comments to the talk page. If you do not want to join the wiki and would still like to comment, feel free to email me.
- Click here to join the wiki.
- Click here to email me.
- Click here to post general comments about the open dissertation.
- Click here to post comments to this chapter's talk page.
A pdf version of the Introduction can be downloaded here. This is the final snapshot of the dissertation that has been approved by my committee. This file will inevitably become out of sync with the wiki pages.
A zip folder containing the LaTeX code can be down loaded here.
Note
This chapter ended up becoming an Open Access paper on PLoS ONE. You can view it on their website, comment if you like and you can cite it below,
Maloney A, Herskowitz LJ, & Koch SJ (2011) Effects of Surface Passivation on Gliding Motility Assays. PLoS ONE, 6(6): e19522. doi: 10.1371/journal.pone.0019522.
Acknowledgements
I would like to thank Dr. Haiqing Liu (while in the lab of Dr. Gabriel A. Montano) for supplying kinesin to our lab. I would also like to thank Dr. Susan Atlas and the support from DTRA CB Basic Research Program under Grant No. HDTRA1-09-1-008 and the UNM IGERT on Integrating Nanotechnology with Cell Biology and Neuroscience NSF Grant DGE-0549500. Finally, I'd like to thank Dr. Erik Schaeffer for his discussions on temperature stabilization.
Introduction

|
| Figure 1: Image of our mascot "Kiney" at the microscope where I perform experiments. The plush doll form of Kiney was made by Child's Own Studios. |
Both kinesin and microtubules are vital components of eukaryotic cells. Kinesin is used as a vehicle to shuttle items from one part of the cell to another and microtubules are the roads that kinesin travel on. Kinesin walks along microtubules in one direction and does so by converting chemical energy, in the form of ATP hydrolysis, to linear motion (Carter 2005, Vale 1997, Hua 1997, Vale 1985, Goldstein 1999). Biophysical observations of motility can be carried out in many different ways, however, the two most prominent observational techniques investigate kinesin and microtubules via the gliding motility assay or, with an optical trap (Block 1990, Yildiz 2004, Gelles 1988). The gliding motility assay (Bohm 1997, Bohm 2000a, Bohm 2000b) is the experimental procedure of interest for this study and a description on how to prepare a gliding motility assay can be found in Chapter 2.
This study aims to show that there are subtle differences in the speeds at which microtubules will glide at depending on the surface passivation used. It also shows that different surface passivations support different lengths of microtubules. This chapter will discuss the subtle differences to the gliding motility assay already outlined in Chapter 2. I will also discuss some of the technological hurdles that were accomplished in order to obtain stable speed data.
Methods and materials
In this section, I will discuss the technological hurdles required to obtain stable data and the subtle differences in the generic gliding motility assay described in Chapter 2.
Temperature stabilization
|
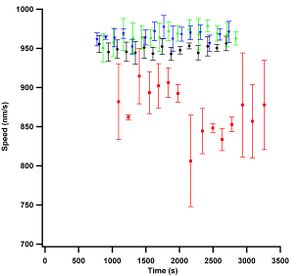
| Figure 2: Graph showing three experiments using different passivation schemes. Note that each individual trace continuously increases in speed over time. |
Temperature stabilization is crucial for observing stable speeds. Without it, I observed microtubule gliding speeds increase over time. Figure 2 shows data that did not use a temperature stabilized objective. The data show a sharp initial increase in speed which, over time would taper but never level off. There are three different assays in the graph, one each of: alpha casein, beta casein and whole casein. A description of the different types of caseins will be given below, briefly they are the constituents of whole casein described in Chapter 2. Using the data from Figure 2, I thought that I observed the goal of this study; which was to observe speed changes dependent on the type of passivation used. However, what I observed was that kinesin is a very sensitive temperature probe as I will discuss in more detail below.
I presented Figure 2 at the Biophysical Society Meeting 2010 where Stefan Diez suggested that the increase in speed over time was due to a temperature effect. I did not initially understand his comment because there was nothing attached to the microscope that could be heating the sample. So, I didn't really do anything about it. I presented more of the same data showing speed changes due to water isotopes and osmolytes at a conference in Santa Fe later that year (Maloney 2010). Dr. Erik Schaeffer of TU Dresden attended the conference and he again told me that there was a temperature issue with my data. He described to me the microscope setup used in his lab which stabilized the temperature of their objective with millikelvin precision. He told me that with this setup, he could tell rather precisely when and if a graduate student in his lab turned on the computer monitor near the microscope. This story blew me away and I of course didn't think it possible but, it most certainly is true as I found out with my experiments. If a computer monitor not even attached to the microscope could register a temperature increase on the objective, then there was a possibility that my microscope was heating up due to the mercury lamp.
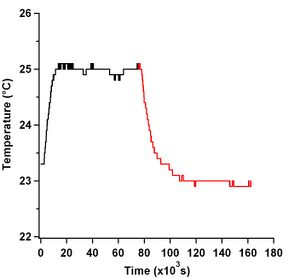
The temperature of the entire microscope does increase over time due to having the mercury lamp on. Figure 3 shows the temperature of the objective due to having the mercury lamp on and shining through it. The temperature of the objective was taken at its top, near where the sample would be. No oil was used on the objective for this measurment. As can be clearly seen, the objective does reach a stable temperature, albeit 5.5 hours after turning the lamp on. This is not ideal for experiments as one would have to wait 5.5 hours before being able to take measurements. This graph does not show the effect of placing a microscope slide at room temperature on the objective as doing so causes the objective temperature to decrease, as can be seen in Figure 4.
|
|
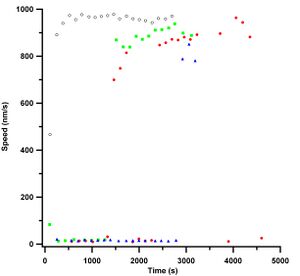
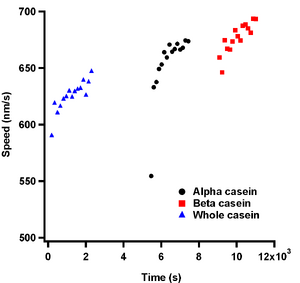
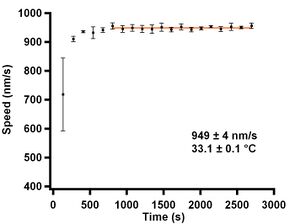
| Figure 5: Graph showing speed values of data from Figure 2 after plotting against the absolute time they were observed. | Figure 6: Graph showing speed values after temperature stabilization. |
Re-plotting the data taken in Figure 2 against absolute time, i.e. from the seconds measured since 1904, shows a different story about microtubule speed measurements and casein passivation than is shown. Thankfully I took all the data in Figure 2 on the same day and when plotted with their time stamps appropriately zeroed from the first image taken, Figure 5, one can clearly see that there is an overall increase in speed as time goes on. One can also see that over the time the slide is on the objective, the microtubule speeds increase. This was due to the fact that the slides were at room temperature and they heated up when on the objective. A plot showing the effect of temperature on the objective for turning the heating element on and off, and adding oil and removing and replacing a slide on the objective can be seen in Figure 4.
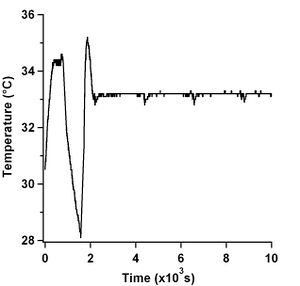
This finding I felt was unfortunate because it basically said that the large differences in gliding speeds was solely due to temperature effects and not due to the chemistry of passivation. Nonetheless, I took this opportunity to build a similar system to what Dr. Schaeffer uses in his lab to stabilize the temperature of my microscope objective (Mahamdeh 2009). As can be seen in Figure 4, with a stable temperature, speed measurements can be recorded with as little as ±4 nm/s errors. This is a feat all to itself since average speeds in the literature can quote errors as large as 50 --- 100 nm/s (Bohm 1997). Figure 4 also shows the large increase in speed observed in my earlier experiments for early times, except now, the data levels off. Figure 4 shows quite nicely that without temperature stabilization, speed measurements would not be stable. Figure 7 shows a movie when there was no temperature stabilization on the objective. The movie shows how the focus would drift which required constant adjustments. The movie loses focus when the overall image gets brighter. This is due to the objective collecting more light from the fluorescent microtubules in solution and not at the surface of the slide where the kinesin is adhered to.
| <html><object width="300" height="300"><param name="movie" value="http://www.youtube.com/v/vq3z-TaDcuI?fs=1&hl=en_US"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/vq3z-TaDcuI?fs=1&hl=en_US" type="application/x-shockwave-flash" allowscriptaccess="always" allowfullscreen="true" width="300" height="300"></embed></object></html> |
| Figure 7: Click here to view the movie via YouTube. Movie showing the focus drift due to temperature instabilities. When the overall image becomes brighter, the focus is drifting. |
A detailed description of the objective heater build can be found in a post done on the website Instructables. Briefly, the objective heater is a PID control circuit using a software interface from TeTech. The control circuitry is connected to two 15 kΩ thermistors. One thermistor controls the heating element and the other is used to measure the objective temperature which is recorded by the software. The heating element used was a Kapton flexible heating element. This design can maintain a temperature to ±0.1°C. There are very few studies that indicate whether or not observation of the gliding motility assay was done with temperature stabilization. It has been shown that temperature does play a crucial role in obtaining stable data (Bohm 2000a, Bohm 2000b) in a few studies, as well as, the ones I have done.
Materials
I like to think of the preparation for all the solutions used in an assay as my mise en place. And, just like no one would ever touch a fellow cook's mise en place in a restaurant, no one but me was allowed to use my solutions. These rules were set in place partly due to how finicky these assays are and how I've learned that keeping a tight control on my solutions allows me to quickly replace chemicals when an assay has gone awry.
I have already spoken about several of the necessary tools used for this assay in Chapter 2. Below, I will link to the tools that do not change and go into greater detail about those that do. The items in the list that do not have links, will be described below.
- PEM buffer
- Antifade & PEM-Glu
- Tubulin for Microtubule polymerization & Taxol for microtubule stabilization.
- Kinesin & PEM-A
- Flow cells
- Surface passivation chemicals
Passivation solutions
This experiment used the different bovine casein constituents to investigate speed variations of microtubules in the gliding motility assay. In the temperature stabilization section of this chapter, my initial experiments saw huge differences in speeds using different caseins. As was already mentioned above, the large speed variations were due to temperature effects and not due to the passivation chemicals. Once I stabilized the temperature of my objective, I continued to investigate if there were any variations in speed due to surface passivation. It turns out that there was a difference in speed measurements due to surface passivation, albeit a more subtle difference than what I saw initially without temperature stabilization. To begin this experiment, I purchased the following caseins all from bovine milk.
- A combination of the αs1- and αs2-caseins purified to 70% (Sigma C6780)
- β-casein purified to 98% (Sigma C6905)
- κ-casein purified to 70% (Sigma C0406)
- Whole casein (Sigma C7078)
Each casein component was reconstituted in PEM under constant stirring. I will outline below the different solutions that were prepared for this experiment giving the naming convention for each solution in the subheading.
W-PEM
For a detailed description of how to make W-PEM, please see Chapter 2.
α-PEM
The subscript s attached to the alpha casein means sensitive since the alpha caseins are the first caseins to be removed from solution by adding over 4 mM calcium. αs1-casein comprises approximately 37% of whole bovine casein while αs2-casein comprises only 10% (Fox 1998a). A mixture of the αs1- and αs2-caseins was purchased from Sigma.
Alpha PEM has 1.0 mg/mL α-casein in PEM. Dissolving α-casein in PEM required approximately 60-80 minutes of constant stirring at room temperature. The procedure I followed to make α-PEM is as follows.
- Weigh out 25 mg of α-casein.
- Add the 25 mg of α-casein to 25 mL of PEM.
- Stir for approximately 60-80 minutes or until there are no longer visible precipitates in solution.
- Allow the foam to settle if any.
- Aliquot into convenient screw top vials.
α-PEM will last for 6 months stored at 4°C. No additional filtering was done on the α-PEM solution.
β-PEM
β-casein comprises approximately 35% of the protein in whole bovine casein. It takes approximately 400 mM calcium to remove it from a solution of skim milk (Fox 1998a).
β-PEM has 1.0 mg/mL β-casein in PEM. The procedure for making β-PEM is very similar to how I made α-PEM except for the length of time required to mix.
- Weigh out 25 mg of β-casein.
- Add the 25 mg of β-casein to 25 mL of PEM.
- Stir for approximately 30-40 minutes or until there are no longer visible precipitates in solution.
- Allow the foam to settle if any.
- Aliquot into convenient screw top vials.
β-PEM will last for 6 months stored at 4°C. No additional filtering was done on the β-PEM solution.
κ-PEM
κ-casein is structurally very different from α and β casein. Kappa casein is a glycoprotein and was depicted in Chapter 2's figure of a casein micelle as the hairs. Kappa casein is thought to stabilize the casein micelle and it is unaffected by calcium concentration. It can even help to stabilize alpha and beta caseins from calcium precipitation (Fox 1998a).
κ-PEM has 1.0 mg/mL κ-casein in PEM. The procedure for making κ-PEM is very similar to how α-PEM is made, except for the length of time required to mix.
- Weigh out 25 mg of κ-casein.
- Add the 25 mg of κ-casein to 25 mL of PEM.
- Stir for approximately 15-20 minutes or until there are no longer visible precipitates in solution.
- Allow the foam to settle if any.
- Aliquot into convenient screw top vials.
κ-PEM will last for 6 months stored at 4°C. No additional filtering was done on the κ-PEM solution.
M-PEM
From the stated amounts of the individual casein components, I mixed a whole casein type solution using α-PEM, β-PEM, and κ-PEM which I called M-PEM. I decided to do this because I wanted to see if I could artificially reproduce the results obtained from W-PEM which used the purchased whole bovine casein.
Since all the components of casein were already in solutions of PEM, I just mixed them in a vial to obtain M-PEM.
- 49% α-casein or 980 μL of α-PEM.
- 37% β-casein or 740 μL of β-PEM.
- 14% κ-casein or 280 μL of κ-PEM.
M-PEM will last for 6 months stored at 4°C. No additional filtering was done on the M-PEM solution.
I did not investigate the solubility of the caseins in a solution of PEM. Casein solubility is a complicated function of other casein constituents in solution (Zittle 1961, Zittle 1963), temperature (Payens 1963, O'Connell 2003, Leclerc 1997), genetic variants (Thompson 1969), ionic strength and types of salts in solution (Thompson 1969), calcium ion concentration (Zittle 1961, Zittle 1963, Thompson 1969), and pH (Bingham 1971).
Experiment and data analysis
The speed at which microtubules glide at in the motility assay depends on a number of factors. One of those factors is the pH of the solution. In my PEM solution, the amount of NaOH is approximate since each solution of PEM is pH-ed to 6.89. Bohm (Bohm 2000b) shows that gliding speed is affected by both the pH and the ionic strength of the solution the motors are in. In an effort to reduce as many variables as possible for speed measurements, I chose to maintain the pH of the PEM buffers at exactly 6.89 and vary the amount of NaOH necessary to achieve this pH.
For a complete description of the gliding motility assay, please see Chapter 2. Briefly, polymerization was carried out in a Thermo PCR Sprint thermocycler held constant at 37°C and incubated for 30 minutes using 1 μL of the 29% rhodamine-labeled and 71% unlabeled tubulin. After 30 minutes, the microtubules were fixed and diluted by 200x by adding 199 μL of PEM-T. Polymerized microtubules were stored at room temperature and protected from ambient light until used in a motility assay. Storing polymerized microtubules at 4°C will cause rapid depolymerization (Shelanski 1973).
Gliding motility assay
Experiments followed the same recipe outlined in Chapter 2. The steps taken for this experiment were as follows.
- Prepared a flow cell.
- Incubated the flow cell with 1.0 mg/mL of the various caseins in PEM for 10 minutes.
- During the 10 minute incubation, kinesin was diluted in 0.5 mg/mL of the same casein in PEM used in Step 2, plus 1 mM ATP. The final concentration of kinesin used in these experiments was 27.5 μg/mL.
- After 10 minutes, kinesin was flowed into the flow cell by fluid exchange. This was then allowed to sit for 5 minutes.
- During the various incubation steps, a new motility solution was prepared for each assay. The motility assays consisted of:
- After the 5 minute incubation with kinesin, the motility solution was introduced to the flow cell by fluid exchange.
- The flow cell was then sealed with nail polish.
In order to not dilute the Taxol used to stabilize microtubules, it is added in the motility solution at the same concentration used in PEM-T. Preventing dilution of chemicals is why 1 mM ATP is added to the motility solution as well. The kinesin home page and Verma et al. (Kinesin home page, Verma 2008), suggest that adding casein to the motility solution enhances the chances of a gliding motility assay to work properly. I observed that adding casein to the motility solution causes microtubules to undergo non ideal motility, i.e. they move in tight circles and end up wrapping around themselves such that the microtubules look like squiggles and are untrackable. In order to prevent such behavior, I did not include casein in the motility solution.
After sealing the flow cell, the slide was immediately placed on the microscope for observation. Data was taken at 5 frames per second with a single exposure time for each frame of 100 ms. The camera software (KochLab) was set to take a total of 600 images for one region of interest (ROI) and had an EMCCD gain of 150. The time to capture 600 frames was approximately 2 minutes of total exposure for one ROI. After the camera had captured 600 frames, I moved the sample on the microscope. I did this because I wanted to get a broad sample of speed measurements from the flow cell. Kinesin may not be distributed uniformly on the surface of the flow cel and may give different speed measurements dependent on where the observation took place. Hunt et al. (Hunt 1994) showed that microtubules would slow down against viscous loads if the kinesin concentration was low. My assays never used small concentrations of kinesin but, to ensure that I had a sampling of speed that did not have to take into account kinesin coverage, I moved the sample around. Each slide was repositioned on the microscope for a total of 15 times giving me 15 ROIs to analyze data from.
Data analysis
Microtubules were then tracked using a custom LabVIEW tracking application written by Larry Herskowitz (KochLab). The tracking software (KochLab) used image segmentation algorithms to identify microtubule ends via pattern matching. Microtubules successfully tracked for fewer than 100 consective images were discarded. This lower bound of images was found through experiment to give reliable data. Microtubules that could be tracked with fewer images did not consistently give high fidelity data and thus a lower bound for images was set. The software was unable to track microtubules that had a segmented area less than 55 pixels due to the pattern matching algorithms used. This put a lower bound on the length of trackable microtubules to be around 2 μm. Finally, tracking of microtubules stopped if the microtubule crossed other microtubules or if it came too close to the edges of the field of view.
Once tracking was complete, the X and Y pixel values for the ends of microtubules were analyzed with another custom LabVIEW analysis program written by Larry Herskowitz and Steve Koch (KochLab). This program calculated the speeds at which microtubules were moving at by subtracting the pixel values of consecutive tracked microtubule images and dividing by the time between frames.
[math]\displaystyle{ s=166.7\left(\frac{nm}{s}\right)\times\frac{\sqrt{(x_{n+1}-x_n)^2+(y_{n+1}-y_n)^2}}{t_{n+1}-t_n} }[/math]
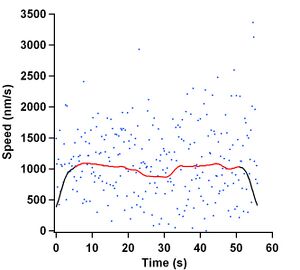
These values are plotted in Figure 8 as the blue markers. As one can see, there is a lot of noise in these raw measurements. I believe that the cause is because microtubule ends are never permanently attached to the kinesin surface and thus undergo Brownian motion transverse to the motility direction. To alleviate this noise, a sliding Gaussian window of 2 seconds was used to smooth the data. This newly smoothed data is depicted as the black curve in Figure 8. Boundary effects due to the finite sample size are quite evident in the graph. There are a lot of techniques that try to compensate for these edge effects, however, we as a group decided to not take them into consideration. Instead, we decided to remove 5 seconds worth of points from the beginning and end of the data set. This completely removed all the edge effects due to smoothing.
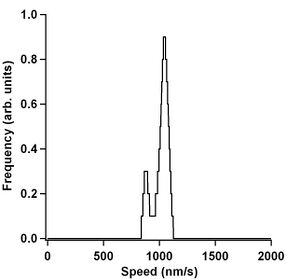
Kernel density estimation (KDE) (Silverman 1986) was then done on all the individual microtubules. Making a KDE is very similar to making a histogram of the speed data. Except that the KDE method applies a Gaussian kernel around every point instead of placing a data point in a bin. The Gaussians are then summed together to give a smooth representation of what the most likely microtubule speed is. Figure 9 shows the KDE of the microtubule data in Figure 8. The bandwidth used in this kernel was 20 nm/s. As you can see in Figure 8, The smoothed red line data is not completely flat. This is because the microtubule does not travel at the same speed all the time. It can pause, stop completely, or even slow down. Using the KDE method allows one to see these subtle differences in speeds and is quite evident in Figure 9.
Speed versus time data for all the microtubules in an individual region of interest (ROI) were then concatenated together (all the smoothed data similar to Figure 8) and the most likely speed for a ROI was extracted using the KDE method with a Gaussian kernel of width 50 nm/s. Using the KDE method instead of a simple mean reduces the sensitivity to microtubule pausing or stalling, which was evident in many assays. We used a large kernel width to reduce sensitivity to possible speed changes due to number of kinesin motors or other causes (Gagliano 2010). The most likely speeds for individual regions were then plotted versus time to determine when the slide had reached thermal equilibrium with the objective, see Figure 6. The initial 5 data points were removed for all data sets indicating that it took about 10 minutes for the slide to reach a stable temperature. Each assay; alpha, beta, kappa, whole, and mixed casein was repeated 3 separate times on different days and with different kinesin aliquots. When possible, the mean and standard error of the mean was computed for the three data points for a given assay time.
Results and discussion
Results
Data was taken at 33.1 ± 0.1°C as measured from the top thermistor on the objective. This temperature is well above the temperature the objective would reach due to long-term heating, see Figure 3, from the Hg lamp and was found to give consistent data, Figure 6. I observed that the closer the ROI was to the boundaries of the flow cell, the slower the microtubule gliding speed was. I also observed that the propensity for depolymerization increased near the boundaries of the flow cell. In order to obtain consistent data and prevent depolymerization of the microtubules, gliding assays were observed in the center of the flow cell channel, except where otherwise noted. All images have had dead pixels removed by an interpolation function and have been false colored using ImageJ's green fire blue LUT using a custom LabVIEW 7.1 application written by Larry Herskowitz (KochLab).
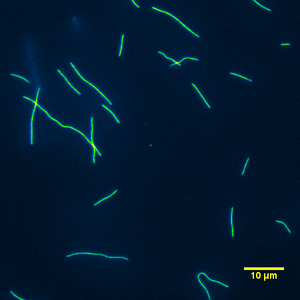
Bovine alpha casein (a mixture of αs1, 37% and αs1, 10%) constitutes approximately 47% of whole bovine casein (Fox 1998a). This passivation was capable of supporting small microtubules and longer ones as can be seen in Figure 10 & 11. When using alpha casein, the gliding motility assay worked every time except when the kinesin or antifade system lost its effectiveness for maintaining a gliding assay.
|
<html><object width="300" height="300"><param name="movie" value="http://www.youtube.com/v/UnRTr1FUdtY?fs=1&hl=en_US"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/UnRTr1FUdtY?fs=1&hl=en_US" type="application/x-shockwave-flash" allowscriptaccess="always" allowfullscreen="true" width="300" height="300"></embed></object></html> |
| Figure 10: Typical image from a gliding motility assay using alpha casein as the surface passivator. | Figure 11: Typical movie from a gliding motility assay using alpha casein as the surface passivator. |
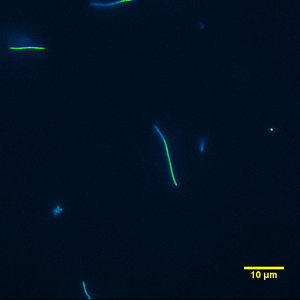
Bovine beta casein comprises approximately 35% of whole casein and visible precipitates took less time to dissolve in PEM as compared to alpha casein. Figure 12 & 13 shows that beta casein as a surface passivator was not ideal. It did not support smaller microtubules and did not in general have very many motile microtubules in any assay. The microtubules that were motile, were typically quite long. Beta casein also caused the microtubule's minus and positive ends to detach from the kinesin surface while undergoing motility more often than the other passivation schemes. This caused errors in the tracking and thus did not give consistent data. This is unfortunate as it can be purified to better than 98% making this component of whole casein the purest commercially available. Another interesting phenomenon observed when using beta casein was that motile microtubules were not found in the center of the channel for the flow cell. Motile microtubules were observed, but they were always found off center to the flow cell channel. I am not sure of the reason for this but, it may depend on kinesin or casein concentration. I did not vary the input kinesin or casein concentration for these studies.
|
<html><object width="300" height="300"><param name="movie" value="http://www.youtube.com/v/6IXpaC_mqZk?fs=1&hl=en_US"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/6IXpaC_mqZk?fs=1&hl=en_US" type="application/x-shockwave-flash" allowscriptaccess="always" allowfullscreen="true" width="300" height="300"></embed></object></html> |
| Figure 12: Typical image from a gliding motility assay using beta casein as the surface passivator. | Figure 13: Typical movie from a gliding motility assay using beta casein as the surface passivator. |
Kappa casein, compared to alpha and beta, is structurally very different. It is a glycoprotein and is thought to stabilize the casein micelle (Fox 1998a, Walstra 1999, Phadungath 2005) by sterically hindering the aggregation of too many casein sub-micelles. It did not support motility in a very consistent manner as can be seen from Figure 14 & 15. As was the case for beta casein, kappa casein did not support motility in the center of the channel of the flow cell. Stuck microtubules were found near the center of the flow cell and near the tape. However, between the boundaries of the flow cells and away from the center of it, there was motility. In the areas that motility existed, kappa casein was able to support motility of long and short microtubules with the exception of extremely short microtubules found to move only in the alpha or whole casein assays. The very short microtubules either remained stuck to the surface or exhibited motility for a very short period of time before going into solution. Kappa casein did a remarkable job of adhering microtubules to the slide much like how poly-L-lysine is used to fix microtubules on a slide (Vater 1995).
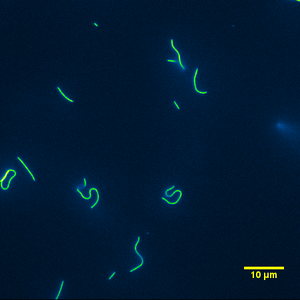
|
<html><object width="300" height="300"><param name="movie" value="http://www.youtube.com/v/s6cATn48z3Y?fs=1&hl=en_US"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/s6cATn48z3Y?fs=1&hl=en_US" type="application/x-shockwave-flash" allowscriptaccess="always" allowfullscreen="true" width="300" height="300"></embed></object></html> |
| Figure 14: Typical image from a gliding motility assay using kappa casein as the surface passivator. | Figure 15: Typical movie from a gliding motility assay using kappa casein as the surface passivator. |
Whole bovine casein is the passivator of choice when doing gliding motility experiments. Whole bovine casein worked remarkably well for sustaining motility, Figure 15 & 17. Similar to the alpha casein passivation, whole casein worked every single time and gave consistent data. It was only when we deemed either the kinesin or the antifade system to have lost its effectiveness at maintaining microtubule gliding that the assay did not work. Of the 5 types of bovine casein solutions tested, whole casein required heat in order for visible precipitate to completely dissolve into a solution of PEM. I did not observe any adverse affects to motility by heating whole casein.
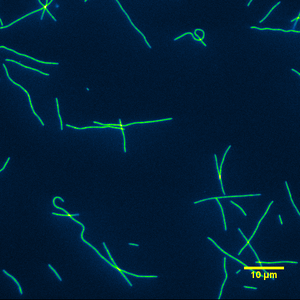
|
<html><object width="300" height="300"><param name="movie" value="http://www.youtube.com/v/LV9ELcgZT6I?fs=1&hl=en_US"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/LV9ELcgZT6I?fs=1&hl=en_US" type="application/x-shockwave-flash" allowscriptaccess="always" allowfullscreen="true" width="300" height="300"></embed></object></html> |
| Figure 16: Typical image from a gliding motility assay using whole casein as the surface passivator. | Figure 17: Typical movie from a gliding motility assay using whole casein as the surface passivator. |
Mixing whole casein from the individual constituents of alpha, beta, and kappa was also used as a surface passivator. The mixed whole casein consisted of 49% alpha casein, 37% beta casein and, 14% kappa casein which is very similar to what Fox and McSweeney state as the casein components of bovine milk (Fox 1998a). Mixing it was easy since each component was already in a PEM solution. The behavior of the mixed bovine whole casein was indistinguishable from the purchased whole casein or the alpha casein passivation, Figure 18 & 19.
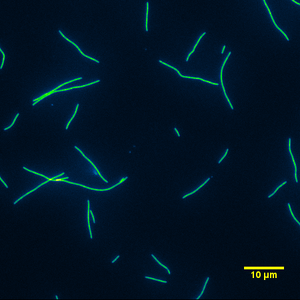
|
<html><object width="300" height="300"><param name="movie" value="http://www.youtube.com/v/C6CjWUu1E0k?fs=1&hl=en_US"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/C6CjWUu1E0k?fs=1&hl=en_US" type="application/x-shockwave-flash" allowscriptaccess="always" allowfullscreen="true" width="300" height="300"></embed></object></html> |
| Figure 18: Typical image from a gliding motility assay using mixed casein as the surface passivator. | Figure 19: Typical movie from a gliding motility assay using mixed casein as the surface passivator. |
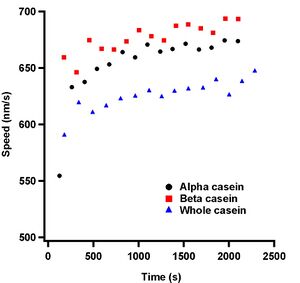
Figure 20 shows the mean speed measurements for 15 different regions of interest for the alpha, beta, whole and mixed casein assays. Each region of interest is the mean and SEM from three separate samples. The passivator that gave the most consistent speed was alpha casein. The mean speed and SEM from our alpha casein measurement was 949 ± 4 nm/s. Purchased whole casein and mixed casein performed remarkably similarly and displayed average speed values of 966 ± 7 nm/s and 966 ± 6 nm/s respectively. Bovine beta casein performed poorly in comparison to alpha, whole, or mixed caseins and we measured the mean speed to be 870 ± 30 nm/s.
Figure 21 shows the observed speeds for kappa casein passivation. Since there were so many areas where no motility was observed in this assay, it was difficult to determine a mean speed measurement for each ROI as was done in Figure 20. However, it does appear that when motile, the speeds were around 870-880 nm/s with kappa casein as the surface passivator. This was similar to how beta casein performed.
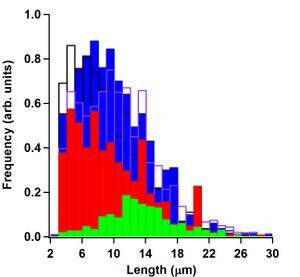
Figure 22 shows a histogram of microtubule lengths. To obtain lengths, an erosion algorithm was used on binary images of only the microtubules that were tracked. The erosion algorithm comes as a standard function in LabVIEW 7.1. The skeleton algorithm, as it is called in LabVIEW, is not optimized for measuring microtubule lengths and tended to overestimate them. Because of this, another algorithm was used on the skeletonized microtubules, namely a Convex Hull perimeter calculation, also found in the LabVIEW library of functions. Dividing the Convex Hull perimeter by 2 gave lengths that consistently fell in between manual measurements done by myself and Koch. As can be seen in the figure, alpha casein (filled blue bars), whole casein (empty purple line bars) and mixed casein (empty black line bars) all were able to sustain motility with smaller microtubules 2 --- 6 μm range. Beta casein (filled green bars) had significantly fewer microtubules that were tracked, however, those that were tended to be longer than the ones tracked in the alpha casein assay. Kappa casein (filled red bars) had many more trackable microtubules than beta casein but significantly fewer than alpha casein. The smallest trackable microtubules using our image segmentation algorithm are around 2 μm. Figure 22 shows that there are relatively few 2 μm microtubules in any of the assays. Kappa casein, however, had no microtubules that fell in the 2 --- 3 μm range but it had a large number of microtubules that were in the 20 --- 21 μm range. Length measurements using this method are not optimized for precision. This method does give a way to see the relative size distribution differences in the assays.
Discussion
It would appear that the more difficult it was to dissolve casein, the better it worked as a surface passivator. This may be a coincidence, or it may relate to the manner in which casein adsorbs to the glass. Whole casein was by far the most difficult to dissolve visible precipitate into PEM without heating. Alpha casein came in second, beta third and kappa fourth in terms of the time to dissolve completely in PEM at room temperature. Whole casein is approximately 50% alpha casein and seeing how well alpha casein performed as a surface passivator, it was not surprising to find that whole casein also performed well. The differences in measured speeds between alpha and whole casein could be a result of how kinesin is supported by the different caseins, or it could be due to differences in surface-coverage for passivation by the casein. It has been shown that using substrates of differing hydrophilicity with whole casein passivation affects the activity of kinesin (Huang 2005).
Purchased whole and mixed casein were practically indistinguishable. They performed almost identically and had the same measured speeds. Mixed whole casein has an upper bound of 20% impurities in it. This amount is very similar to the amount of impurities alpha casein has in it yet, mixed whole casein performed exactly how purchased whole casein did. This similarity between mixed and purchased whole casein suggests that the speed difference between alpha and whole casein is not due to impurities but rather that it is due to how kinesin is supported by the casein micelles or, how casein interacts with the glass. To elucidate this effect, it would be prudent to measure the speeds for the various caseins as a function of casein and kinesin concentrations during incubation. I have not performed these experiments.
Beta casein would have been the most ideal protein to use as the surface passivator since it can be purified to greater than 98% purity. Higher protein purity is advantageous for systematically producing devices that use kinesin and microtubules as sensors. However, beta casein did not perform very well. It had the least number of motile microtubules to track, it was not very reliable, and had a large distribution in speeds. It is possible, though, that varying other parameters such as kinesin concentration, or beta casein concentration could restore reliable motility. Again, these are future experiments.
Kappa casein would be an attractive surface passivator just for its ease in dissolving in PEM. However, where motility exists in the flow cell is not consistent and it never occurs in the center of the channel where I have observed the most consistent speed measurements from other assays. With kappa casein, many long and stable microtubules permanently stuck to the surface. I am not sure if the sticking is caused from kinesin attaching to microtubules and then somehow being impeded from moving or, if there is actually no kinesin on the kappa casein surface and the microtubules are just attracted to the kappa casein or glass.
Of the five types of bovine caseins used to observe the gliding motility assay, alpha casein performed very well. It is the easiest of the three commercially available bovine casein constituents to dissolve in PEM and can be purchased at 70% purity. Most likely, the 30% contaminants are from other casein components. It performed well every time an assay was prepared and worked just as well as mixed and purchased whole casein.
Conclusion
There are a wide variety of surface passivation strategies in the literature. This study showed that there are differences in how the gliding motility assay is observed dependent on the type of passivaiton used. Bovine alpha casein was the most reliable passivator when following the most typical gliding assay protocol found in [http://www.openwetware.org/wiki/User:Andy_Maloney/Kinesin_%26_Microtubule_Page Chapter 2. This is why I have chosen to use it for my following studies using isotopes of water in the gliding motility assay.
Future work
There are a great number of experiments that could be done to further this line of research. For instance, varying the amount of kinesin used in each assay could illuminate more characteristics of how kinesin interacts with the surface passivation. Also, varying the casein constituent concentration used to passivate the surface could bring to light the possibility that either beta or kappa casein is a better surface passivator than alpha casein. And finally, since the individual constituents of bovine casein can be purchased separately, one can devise experiments that uses engineered whole casein as the surface passivation. One could even use dephosphorylated alpha or beta casein as components to the engineered surface passivation.
Return to the table of contents.
References
- Alberty, R. A. (1968). Effect of pH and metal ion concentration on the equilibrium hydrolysis of adenosine triphosphate to adenosine diphosphate. The Journal of Biological Chemistry, 243(7), 1337-1343.
- Berliner, E., Young, E., Anderson, K., Mahtani, H., & Gelles, J. (1995). Failure of a single-headed kinesin to track parallel to microtubule protofilaments. Letters to Nature, 373(23), 718-721. doi: 10.1038/373718a0.
- Bingham, E. W. (1971). Influence of temperature and pH on the solubility of αs1-, β- and κ-casein. Journal of Dairy Science, 54(7), 1077-1080.
- Block, SM, Goldstein, LSB, & Schnapp, BJ (1990). Bead movement by single kinesin molecules studied with optical tweezers. Nature, 348, 348-352. doi: 10.1038/348348a0.
- Bohm, K. J., Steinmetzer, P., Daniel, A., Baum, M., Vater, W., & Unger, E. (1997). Kinesin-driven microtubule motility in the presence of alkaline-earth metal ions: indication for a calcium ion-dependent motility. Cell motility and the cytoskeleton, 37(3), 226-31.
- Bohm, K. J., Stracke, R., Baum, M., Zieren, M., & Unger, E. (2000). Effect of temperature on kinesin driven microtubule gliding and kinesin ATPase activity. FEBS letters, 466(1), 59-62. doi: 10.1016/S0014-5793(99)01757-3.
- Bohm, K. J., Stracke, R., & Unger, E. (2000). Speeding up kinesin-driven microtubule gliding in vitro by variation of cofactor composition and physicochemical parameters. Cell biology international, 24(6), 335-41. doi: 10.1006/cbir.1999.0515.
- Carter, N. J., & Cross, R. A. (2005). Mechanics of the kinesin step. Nature, 435(7040),308-12. doi: 10.1038/nature03528.
- Fox, P., & McSweeney, P. (1998). Chapter 4: Milk Proteins. Dairy Chemistry and Biochemistry (1st ed., pp. 146-238). London: Blackie Academic & Professional.
- Fox, P., & McSweeney, P. (1998). Chapter 9: Heat-induced changes in milk. Dairy Chemistry and Biochemistry (1st ed., pp. 347-378). London: Blackie Academic & Professional.
- Gagliano, J., Walb, M., Blaker, B., Macosko, J. C., & Holzwarth, G. (2010). Kinesin velocity increases with the number of motors pulling against viscoelastic drag. European biophysics journal: EBJ, 39(5), 801-13. doi: 10.1007/s00249-009-0560-8.
- Gelles, J., Schnapp, B. J., & Sheetz, M. P. (1988). Tracking kinesin-driven movements with nanometre scale precision. Nature, 331(6155), 450-3. doi: 10.1038/331450a0.
- Goldstein, L. S. B., & Philp, A. V. (1999). The road less traveled: emerging principles of kinesin motor utilization. Annual review of cell and developmental biology, 15, 141-183.
- Hua, W., Young, E., Fleming, M., & Gelles, J. (1997). Coupling of kinesin steps to ATP hydrolysis. Nature, 388, 390-393.
- Huang, Y., Uppalapati, M., Hancock, W. O. & Jackson, T. N. (2005). Microfabricated capped channels for biomolecular motor-based transport. IEEE Transactions on Advanced Packaging, 28(4), 564-570. doi: 10.1109/TADVP.2005.858330.
- Hunt, A., Gittes, F., & Howard, J. (1994). The force exerted by a single kinesin molecule against a viscous load. Biophysical journal, 67(2), 766-781.
- (Kinesin home page) Kinesin home page http://www.cellbio.duke.edu/kinesin/.
- (KochLab) Links to software used in this thesis. http://kochlab.org/files/Software/Microtubule%20software/.
- Leclerc, E., & Calmettes, P. (1997). Interactions in micellar solutions of β-casein. Physical Review Letters, 78(1), 150-153. doi: 10.1103/PhysRevLett.78.150.
- Mahamdeh, M., & Schaffer, E. (2009). Optical tweezers with millikelvin precision of temperature-controlled objectives and base-pair resolution. Optics Express, 17(19), 17190. doi: 10.1364/OE.17.017190.
- Maloney, A., Herskowitz, L. J., & Koch, S. (Corrected) (2010) Speed effects in gliding motility assays due to surface passivation, water isotope, and osmotic stress. Available from Nature Precedings.
- O'Connell, J. E., Grinberg, V. Y., & de Kruif, C. G. (2003). Association behavior of β-casein. Journal of Colloid and Interface Science, 258(1), 33-39. doi: 10.1016/S0021-9797(02)00066-8.
- Payens, T. A. J., & van Markwijk, B. W. (1963). Some features of the association of β-casein. Biochimica et Biophysica Acta, 71, 517-530.
- Phadungath, C. (2005). Casein micelle structure: a concise review. Songklanakarin Journal of Science and Technology, 27(1), 201-212.
- Shelanski, M. L., Gaskin, F., & Cantor, C. R. (1973). Microtubule assembly in the absence of added nucleotides. Proceedings of the National Academy of Sciences of the United States of America, 70(3), 765-8.
- Silverman, B. W. (1986). Density Estimation for Statistics and Data Analysis. Chapman and Hall: London.
- Thompson, M. P., Gordon, W. G., Boswell, R. T., & Farrell Jr., H. M. (1969). Solubility solvation, and stabilization of αs1- and β-caseins. Journal of Dairy Science, 52(8), 1166-1173. doi: 10.3168/jds.S0022-0302(69)86719-6.
- Vale, R. D., Reese, T., & Sheetz, M. (1985). Identification of a novel force generating protein, kinesin, involved in microtubule-based motility. Cell, 42(1), 39-50.
- Vale, R. D., & Fletterick, R. J. (1997). The design plan of kinesin motors. Annual review of cell and developmental biology, 13, 745-77. doi: 10.1146/annurev.cellbio.13.1.745.
- Vater, W., Fritzsche, W., Schaper, Bohm, K. J., Unger, E., & Jovin, T. M. (1995). Scanning force microscopy of microtubules and polymorphic tubulin assemblies in air and in liquid. Journal of cell science, 108 (Pt 3), 1063-9.
- Verma, V., Hancock, W. O., & Catchmark, J. M. (2008). The role of casein in supporting the operation of surface bound kinesin. Journal of biological engineering, 2, 14. doi: 10.1186/1754-1611-2-14.
- Walstra, P. (1999). Casein sub-micelles: do they exist?. International Dairy Journal, 9(3-6), 189-192. doi: 10.1016/S0958-6946(99)00059-X.
- Yildiz, A., Tomishige, M., Vale, R. D., & Selvin, P. R. (2004). Kinesin walks hand-over-hand. Science (New York, N.Y.), 303(5658), 676-8. doi: 10.1126/science.1093753.
- Zittle, C. A. (1961). Stabilization of calcium-sensitive (α2) casein by kappa Casein: Effect of chymotrypsin and heat on kappa-casein. Journal of Dairy Science, 44(11), 2101-2103. doi: 10.3168/jds.S0022-0302(61)90026-1.
- Zittle, C. A, & Walter, M. (1963). Stabilization of β-casein by κ-casein against precipitation by calcium chloride. Journal of Dairy Science, 46(11), 1189-1191. doi: 10.3168/jds.S0022-0302(63)89243-7.
All of my raw observations, raw data taking, and silly ramblings are contained in my notebook entries. They are the unedited versions of this page.