IGEM:IMPERIAL/2006/project/Oscillator/project browser/Full System/Modelling
From OpenWetWare
Jump to navigationJump to search
| Super Parts | Not applicable | |
|---|---|---|
| Actual Part | Full System logo | |
| Sub Parts | Prey Molecule Generator | Predator Molecule Generator |
Model description of the Molecular Predation Oscillator
|
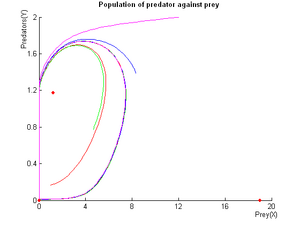
This system of ODEs describes the full system in a chemostat.
|
 |
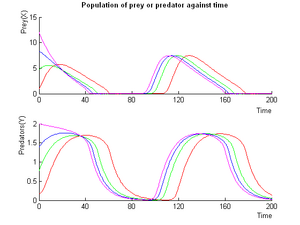 | |
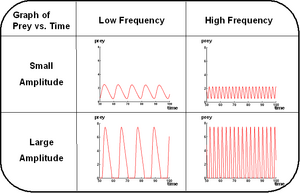
A detailed theoretical study and simulations have shown that the properties of this system are able to fulfill our specifications:
|
 |
Model variables and parameters
| Variables | ||||
|---|---|---|---|---|
| Name | Description | Initial Value | Confidence | Reference |
| AHL | homoserine lactone acting as the prey-molecule | 0 | depends how good is the control of the prey positive feedback. Should be measured. | links |
| luxR | molecule acting as the sensing module for the predator generator | 0 | to be measured as we might have to deal with some leakage of the promoter | links |
| aiiA | molecule acting as the killing module of the prey molecule for the predator generator | to be measured | to be measured as we might have to deal with some leakage of the promoter | links |
| Parameters | ||||
|---|---|---|---|---|
| Name | Description | Value | Confidence | Reference |
| a | maximum synthesis rate of the pLux promoter | to be characterized | to be measured | links |
| a0 | dissociation constant | to be characterized | to be measured | links |
| b | catalysis rate of the AHL-lactonase(aiiA) | variable | to be measured/can be varied by chemostat | links |
| b0 | Michaelis constant for the AHL-lactonase(aiiA) | to be characterized | to be measured | links |
| c | maximum synthesis rate of the pLux promoter | to be characterized | to be measured | links |
| c0 | dissociation constant | variable | to be measured/can be varied by chemostat | links |
| dg | growth dilution due to chemostat wash-out | to be characterized | to be measured | links |
Full derivation of the above equations.
SBML Model
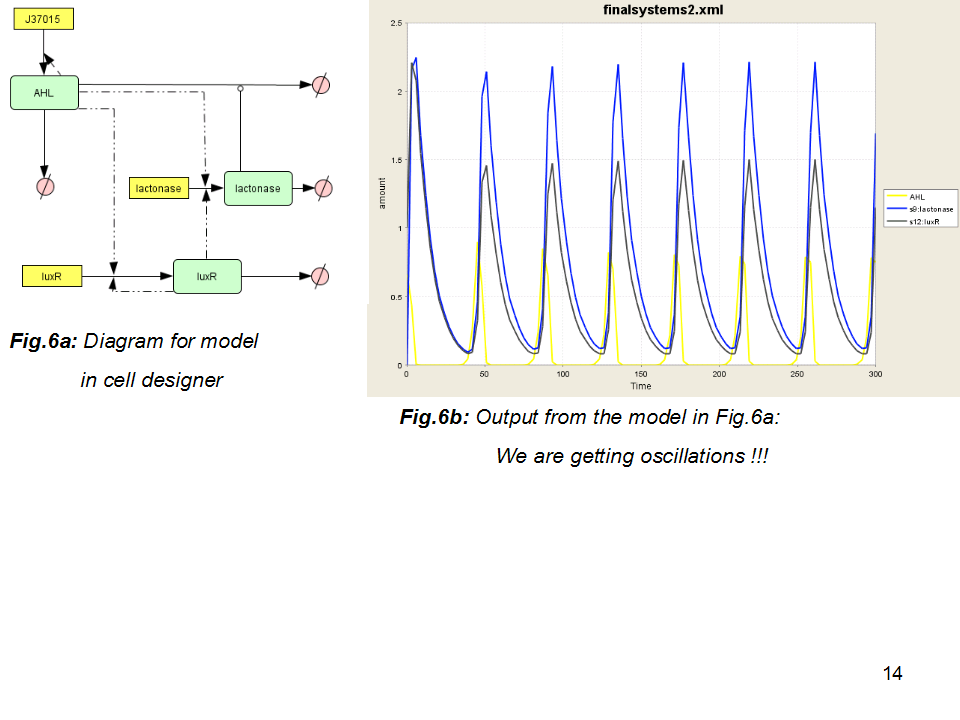
Model assumptions and relevance
- General assumptions on gene expression modelling:
- Quasi-steady state hypothesis on mRNA expression.
- Gene activation can be approximated by Hill equations.
- Assumption on the Chemostat:
- It assumes that the prey molecule generator and the predator molecule generator populations are stable (the cell populations have reached steady-state).
- the degradation of the molecules is mainly due to the wash-out of the chemostat.
- Assumptions linked to the quorum sensing:
- As a first approximation, we assume that luxR and AHL molecules form a heterodimer (even if it has been found that the complex formed is more complicated)
- The concentration of the heterodimer is in equilibrium with the concentration of AHL
- LuxR is constitutively produced and reaches steady state before AHL production begins. [LuxR] in the prey can be considered constant
- The degradation rate of luxR and AHL-lactonase is due to the growth dilution which, in this case, is controlled by the chemostat
- AHL is diffusing freely throughout the system
Characterization
- Characterization of the parameters a, ao shall be done using the Prey Generator Test Contstruct
- Characterization of the parameters c, c0 shall be done using the Predator Generator Sensing Test Construct
- Characterization of the parameters b, b0 shall be done using the Predator Generator Killing Test Construct
- The growth dilution can be controlled by the chemostat washout.
<html> <script type="text/javascript" language="javascript"> var sc_project=1999441; var sc_invisible=1; var sc_partition=18; var sc_security="18996820"; </script>
<script type="text/javascript" language="javascript" src="http://www.statcounter.com/counter/frames.js"></script><noscript><a href="http://www.statcounter.com/" target="_blank"><img src="http://c19.statcounter.com/counter.php?sc_project=1999441&java=0&security=18996820&invisible=1" alt="website statistics" border="0"></a> </noscript> </html>